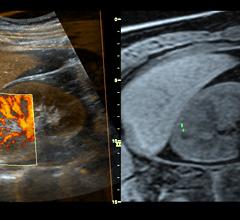
September 8, 2008 - Excluding cardiac causes in patients presenting to the emergency department (ED) with chest pain using myocardial contrast echocardiography (MCE) can prevent unnecessary admissions, making it a cost-efficient tool in the evaluation of these patients, according to a study published in the Sept. 15 issue of the American Journal of Cardiology.
September 8, 2008 - Royal Philips Electronics reached an agreement to acquire India-based Alpha X-Ray Technologies, a ...
Vascular Solutions will partner with Radius Medical Technologies for the U.S. launch of the MICRO Elite Snare, a device ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
September 5, 2008 - Digirad Corp. today announced the initial clinical trial of its new SPECT imaging system ...
September 4, 2008 - GE Healthcare's Diagnostic Cardiology Service team prides itself on exceeding the expectations of ...
Cardiac Science launched its HeartCentrix 6.2 data management product for the physician office to connect Burdick and ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
September 5, 2008 - The results of the SYNTAX study, a head-to-head comparison between drug-eluting stents (DES) using ...
September 5, 2008 - GE Healthcare today said it launched a new ultrasound system for radiology and vascular applications, the LOGIQ E9, which fuses ultrasound images with images from other imaging modalities like CT and MR.
September 2, 2008 - GE Healthcare launched a new ultrasound system for radiology and vascular applications that fuses ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Biotronik GmbH launched the Lumax 540 series, which includes implantable cardioverter defibrillators (ICDs) and a cardiac resynchronization therapy defibrillator (CRT-D), which is used with Biotronik Home Monitoring to enable continuous automatic daily data transmission of the patient’s cardiovascular status via the Internet.
September 5, 2008 - Pathway Medical Technologies Inc., today received FDA clearance to market Jetstream, a peripheral atherectomy catheter designed for use in the treatment of peripheral artery disease (PAD) in the lower limbs.
September 5, 2008 - Boston’s Brigham & Women’s (B&W) Hospital, a 777-bed teaching affiliate of Harvard Medical School ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
September 4, 2008 – St. Elizabeth, in Cincinnati, OH, which serves more than 300 stroke patients annually, is the first ...
The FDA has cleared Medtronic’s Attain StarFix OTW (over-the-wire) lead (Model 4195), an active fixation left-heart lead for cardiac resynchronization therapy (CRT).
The Xcelera R2.2 upgrade is a new version of the Xcelera multimodality cardiology image management, analysis and reporting solution, designed to offer enhanced functionality and greater workflow efficiency.

 September 07, 2008
September 07, 2008