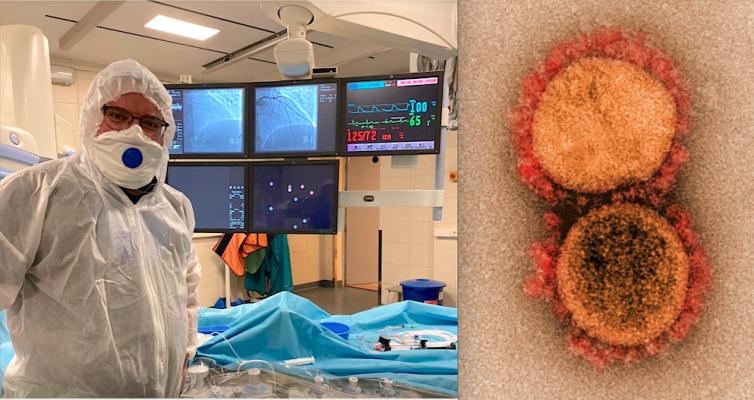
Russian interventional cardiologist Alexey Pankov in full personal protective equipment (PPE) for a cath lab procedure in Moscow during the COVID-19 era. Right, an image of the COVID-19 virus from the National Institutes of Health (NIH). Novel coronavirus has turned out to have a sizable amount of cardiovascular involvement.
It was originally thought novel coronavirus (COVID-19, SARS-CoV-2) was primarily a respiratory disorder, but as larger numbers of patients contracted the virus, it quickly became clear it has many physiological manifestations. The impact of COVID-19 goes well beyond the lungs to impact the cardiovascular system and cause complications in the kidneys, brain and other organs, and critical patients often require care from a multidisciplinary care team.
This article offers an overview of cardiac and vascular complications of COVID-19 observed in the first six months of treating the new virus since the original outbreak in China, first reported in December 2019.
"Our understanding of this disease is changing every week and will certainly change again in the weeks and months to come," explained Vikramjit Mukherjee, M.D., director of medical ICU and the special pathogens program at Bellevue Hospital Center in New York City, who spoke in COVID-19 sessions during the Society for Cardiovascular Angiography and Interventions (SCAI) 2020 virtual conference in May.
At many centers, cardiology was called on to help with some COVID patients. COVID patients with comorbidities were much more likely to have severe symptoms requiring hospitalization, but patients with pre-existing cardiovascular diseases suffer the highest mortality rate of all COVID patients. In some cases, COVID causes acute heart failure, myocarditis, shock and thromboembolism, and the drugs being used to treat the virus can compound cardiac issues by causing arrhythmias, explained Ajay J. Kirtane, M.D., associate professor of medicine at Columbia University Irving Medical Center and director of the cardiac catheterization laboratories at NewYork-Presbyterian Hospital, who also spoke at SCAI.
“We are literally learning as we go along. One of the problems is that COVID is a shape shifter. Just when you think you understand what you are dealing with it changes,” said David Baran, M.D., director for advanced heart failure and transplantation, Sentara Health System, Virgina, who also spoke at SCAI.
ACC Lists the Cardiac Implications of COVID-19
The American College of Cardiology (ACC) listed the following cardiac implications from Chinese COVID case reports in late February 2020.[1] All of these points have since been confiremd in American and European case reports:
• Patients with underlying conditions are at higher risk for complications or mortality from COVID-19 and up to 50 percent of hospitalized patients have a chronic medical illness.
• 40 percent of COVID-19 patients have cardiovascular or cerebrovascular disease.
• 16.7 percent of patients developed arrhythmia.
• 7.2 percent developed acute cardiac injury.
• 8.7 percent of patients developed shock; 3.6% developed acute kidney injury.
• Rates of complication were universally higher for ICU patients.
• Some COVID-19 patients develop myocarditis.
• Fatality rates for comorbid patients are higher than the average population.[2]
- Cancer: 5.6%
- Hypertension: 6%
- Chronic respiratory disease: 6.3%
- Diabetes: 7.3%
- Cardiovascular disease: 10.5%
The novel coronavirus (COVID-19) imaged by the National Institutes of Health (NIH).
"We are seeing significant cardiac involvement, but it is important to note that many of our patients with underlying cardiovascular disease, notably heart failure and coronary disease, are developing significant effects form COVID-19 that is destabilizing conditions, and that presents unique challenge," Justin Fried, M.D., attending cardiologist and heart failure specialist at Columbia University Irving Medical Center, New York City, and assistant professor at at Columbia University and lead author of a report on cardiac involvement in COVID-19 patients.[3]
In patients presenting with what appears to be a typical cardiac syndrome, COVID-19 infection should be in the differential during the current pandemic, even in the absence of fever or cough, Fried said, based on experience from Columbia.
ICU Experience with COVID-19 Patients
In general, COVID patients who went on to require cardiology support usually followed a path of being admitted to the floor, then experience worsening hypoxia requiring admission into the ICU, followed by ARS and the onset of cytokine storm, Mukherjee stated. He said this can lead to renal failure and requires ongoing ventilator and renal replacement therapy (RRT) support, or death.
Mukherjee said there is a lot of learning how to best treat patients as you go along in day-to-day operations because there are no randomized, controlled studies and some of the issues they are facing have never seen before in patients.
 “We still don’t know how to medicate patients to treat cytokine storm,” he stated. “We want to emphasize these are not data-driven therapies.”
“We still don’t know how to medicate patients to treat cytokine storm,” he stated. “We want to emphasize these are not data-driven therapies.”
He explained there is a surge in inflammatory markers in these patients, including D-dimer and troponin. “We had extremely high D-dimers and almost all of our ICU COVID patients had acute respiratory distress syndrome (ARDS). The death rate for the ICU was also much higher than what we are used to,” Mukherjee said.
He said 72 percent of their ICU patients presented with a state of shock and 55 percent patients did not survive. They also saw much higher incidence of acute kidney injury than what was reported from China.
In some of the shock patients, they used VV-ECMO. ECMO was used in five patients, and one died.
What is a Cytokine Storm?
Many severe COVID-19 reactions can be traced to a severe immune reaction in which the body releases too many cytokines into the blood too quickly, known as a cytokine release syndrome, or cytokine storm. The rapid release can be catastrophic to the patient, leading to multiple organ failure. It is believed many of the worse cases of COVID-19 in patients with a multitude of complications are directly related to these cytokine storms. This includes possibly causes of ischemic strokes, shock and organ failure.
Mukherjee said cytokine storm was universal in COVID patients after a few days in the ICU, which led to tachycardia, tachypnea and diaphoresis and elevated D-dimer. Many patients progressed to renal failure, with about 30 percent requiring renal replacement therapy (RRT).
Severe Clotting and Venous Thrombo-emolism (VTE)
Based on reports that demonstrated a strong association between elevated D-dimer levels and poor prognosis, concerns have risen about thrombotic complications in patients with COVID-19. The National Institute for Public Health of the Netherlands asked a group of radiology and vascular medicine experts to provide guidance for the imaging workup and treatment of these complications.[4] The report suggests respiratory failure in COVID-19 is not driven by the development of the acute respiratory distress syndrome alone, but that microvascular thrombotic processes may play a role.
This may have important consequences for the diagnostic and therapeutic management of these patients. There is a strong association between D-dimer levels, disease progression and chest CT features suggesting venous thrombosis. In addition, various studies in patients with COVID-19 have shown a very strong association between increased D-dimer levels and severe disease/poor prognosis.
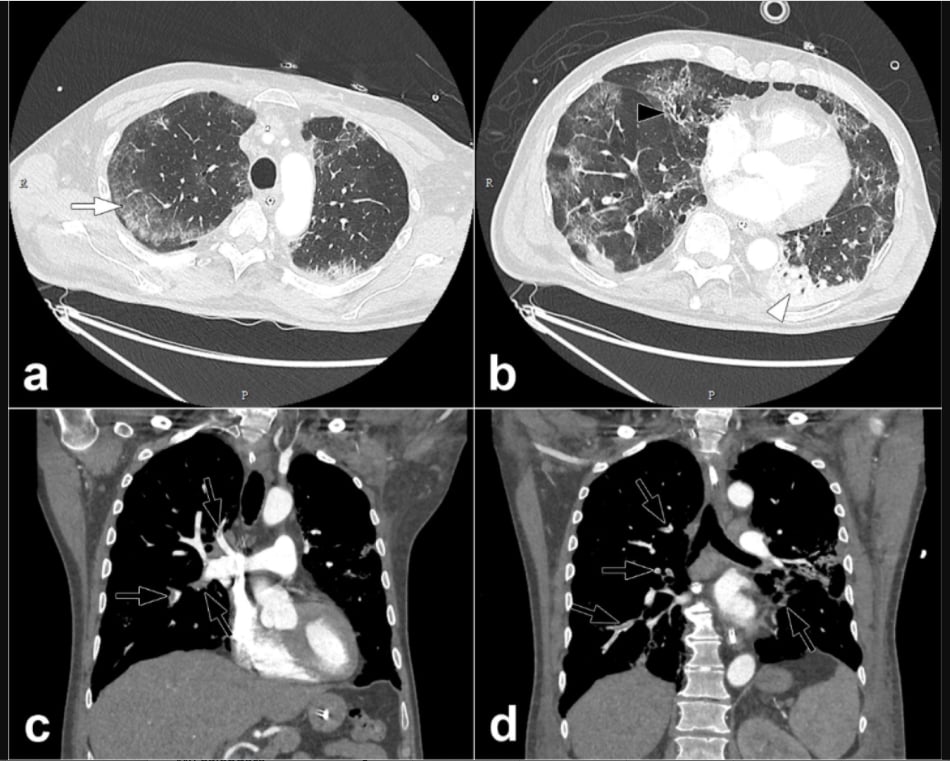
Pulmonary CT angiography of a 68 year old male. The CT scan was obtained 10 days after the onset of COVID-19 symptoms and on the day the patient was transferred to the intensive care unit. Axial CT images (lung windows) (a,b) show peripheral ground-glass opacities (arrow) associated with areas of consolidation in dependent portions of the lung (arrowheads). Interlobular reticulations, bronchiectasis (black arrow) and lung architectural distortion are present. Involvement of the lung volume was estimated to be between 25% and 50%. Coronal CT reformations (mediastinum windows) (c,d) show bilateral lobar and segmental pulmonary embolism (black arrows). Courtesy of RSNA
COVID-19 has since been found in numerous studies to cause hypercoagulability, likely due to the body reacting to severe inflammatory response. Mukherjee said he saw this is about 25-30 percent of his patients. They treated these patients with heparin and Lovenox.
The highly infectious nature of COVID means they try not to move infected patients through hospital corridors to radiology for computed tomography (CT) scans to monitor venous thromboembolism (VTE). Instead, his hospital uses D-dimer as a surrogate for clot formation and used a lot of point-of-care ultrasound (POCUS) to image for clots.
"Left sided cariogenic shock in COVID patients is rare, but there is a substantial amount of right ventricular involvement, and there appears to be a component of micro-emboli," said William O'Neill, M.D., medical director of the Center for Structural Heart Disease at Henry Ford Hospital in early April. "When these patients get really ill, they develop this intense inflammatory response and become pro-thrombotic, so they become prothrombotic and they get spontaneous occlusions of distal extremities, and probably micro-pulmonary emboli, causing pulmonary hypertension."
Pulmonary embolism (PE) is being observed in COVID patients due to this clotting phenomenon. “Acute pulmonary embolism is clearly being recognized as a life-threatening manifestation of COVID-19," said Amir Kaki, M.D., an interventional cardiologist and director of mechanical circulatory support at Ascension St. John Hospital in Detroit.
The U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) June 1, allowing the Abiomed Impella RP catheter-based heart pump to be used in COVID-19 patients with PE and right heart failure.
"Impella RP is an important tool to help cardiologists save lives during this pandemic. As we have demonstrated in our series of patients, early recognition of right ventricular dysfunction and early placement of the Impella RP for patients who are hypotensive can be life-saving,” Kaki explained.
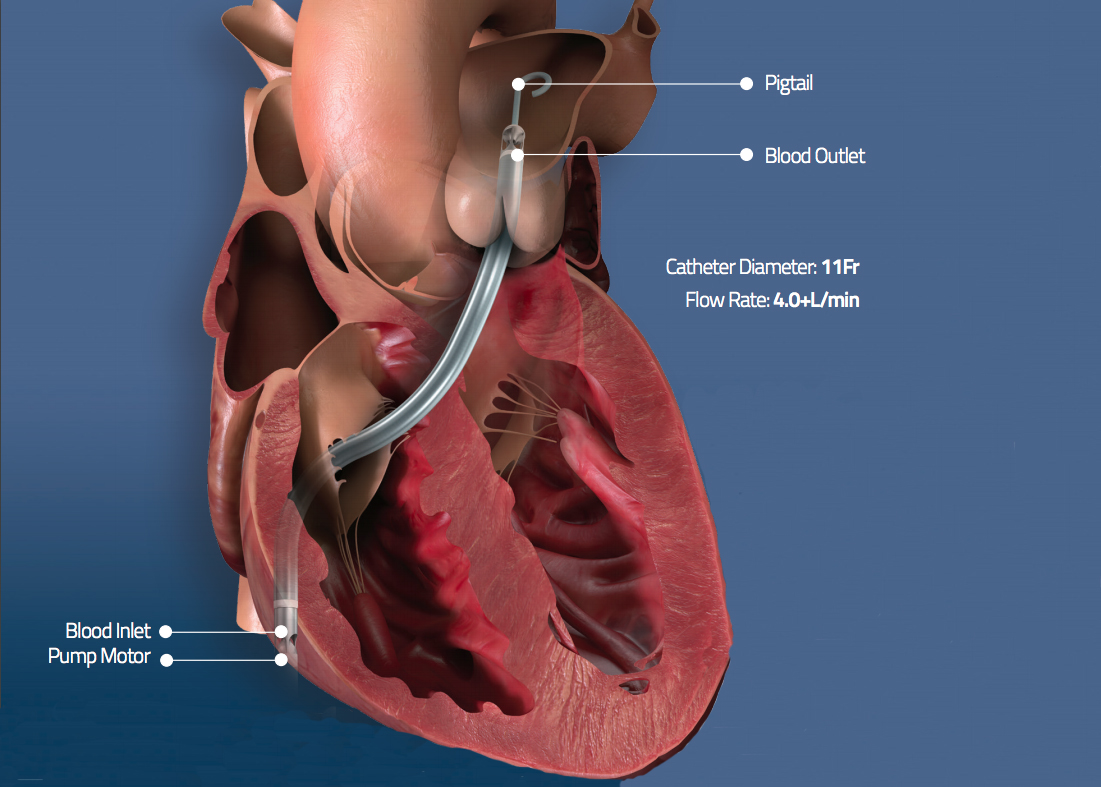
The U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) June 1, allowing the Abiomed Impella RP catheter-based heart pump to be used in COVID-19 patients with PE and right heart failure.
COVID-19 Can Mimic STEMI Heart Attacks
One mystery of COVID is why it sometimes presents as a ST-elevation myocardial infarction (STEMI). ECGs in numerous COVID-19 patients clearly showed a STEMI, requiring immediate revascularization. However, the angiogram in the cath lab showed no blockages, so there was nothing to revascularize.
With the new data module, American College of Cardiology (ACC) began collecting COVID-19 information in May through its National Cardiovascular Data Registry (NCDR) Chest Pain-MI and CathPCI registries. The ACC said the goal is to capture the relationship between COVID-19 and heart disease and maximize data on the cardiac impact of the virus.
SCAI and the Canadian Association of Interventional Cardiology (CAIC) announced in early April the formation of the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI). This effort is recording data from COVID-19 patients who present with STEMI. The registry includes all COVID-19 positive patients or persons under investigation (PUI) with ST-segment elevation or new-onset left bundle branch block (LBBB) with a clinical correlate of myocardial ischemia (including chest pain, dyspnea, cardiac arrest, hemodynamic instability).
COVID-related Arrhythmias and QT-prolongation
The combination of COVID-cause myocarditis along with two of the most common front line drugs used to treat COVID patients can cause life-threatening arrhythmias. Hydroxychloroquine, chloroquine and azithromycin all can cause QT prolongation. These drugs were used prolifically March through May to treat COVID patients, and several cardiology societies warned the first week of April in a joint statement there would likely be a rapid uptick in arrhythmia complications and possible deaths.[5]
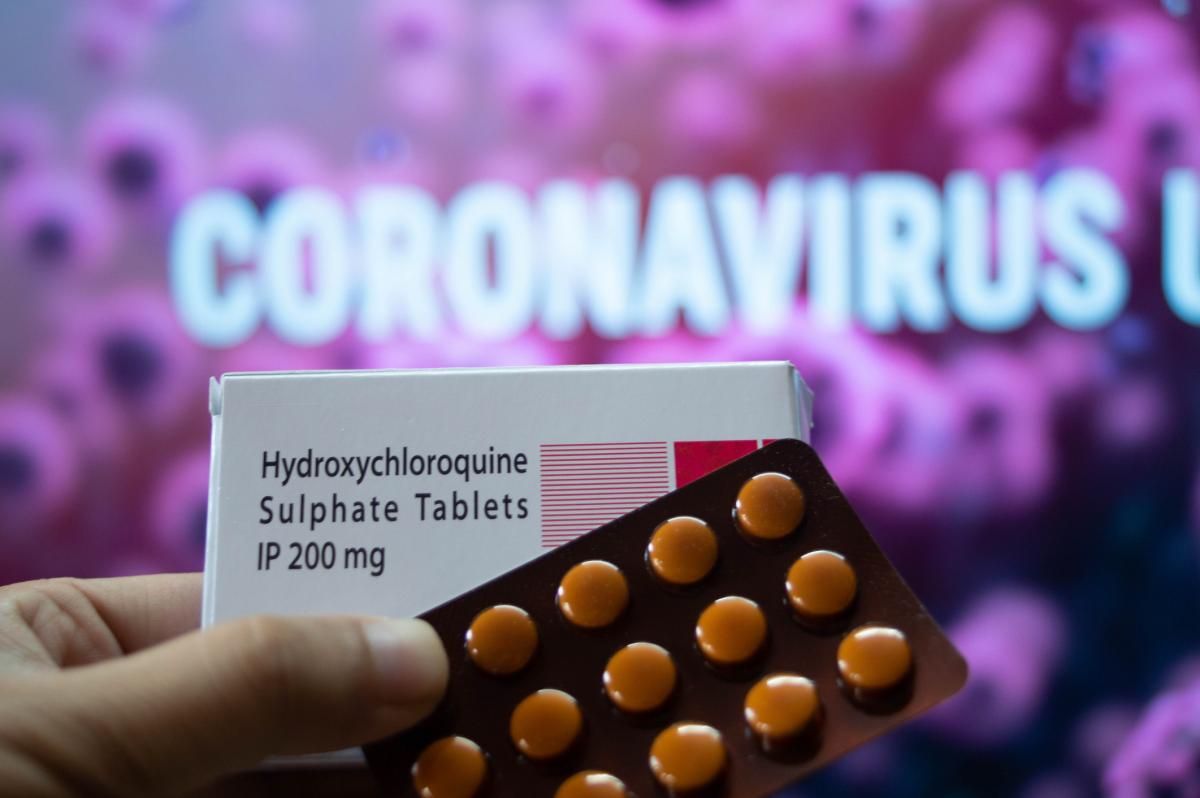 Not surprisingly, the three weeks later in late April, FDA announced it received numerous adverse event reports, including deaths, being filed related to these drugs being used to manage COVID patients. The FDA warned doctors there are serious potentially life-threatening heart rhythm side effects when using hydroxychloroquine and chloroquine in the off-label use to treat COVID-19 patients.
Not surprisingly, the three weeks later in late April, FDA announced it received numerous adverse event reports, including deaths, being filed related to these drugs being used to manage COVID patients. The FDA warned doctors there are serious potentially life-threatening heart rhythm side effects when using hydroxychloroquine and chloroquine in the off-label use to treat COVID-19 patients.
"It is very pertinent because the medications that are being advocated to treat the virus do have specific cardiac considerations we need to keep in mind," Fried explained, who treated at least one COVID patient who suffered from the effects of QT-prolongation.
"All treatments have some potential downside, and both hydroxychloroquine and azithromycin are drugs we tell people not to take all the time in my business. But, in desperate times, people will take measures they feel are necessary to help patients," said Andrew D. Krahn, M.D., FHRS, head of the division of cardiology at St. Paul’s Hospital, and professor of medicine at the University of British Columbia, and vice president of the Heart Rhythm Society (HRS). He is an expert in long QT syndrome (LQTS).
He said it is the combination of underlying heart problems, the drugs bring used for COVID and possibly low potassium in patients that combine to cause severe arrhythmias. He said patients' ECG need to be closely monitored.
Watch Krahn discuss more in the VIDEO: Why QT-prolongation Occurs in COVID-19 Patients on Hydroxychloroquine and Azithromycin.
ECMO or Other Hemodynamic Support for COVID-19 Patients
Cardiology is called in some of the most severe COVID cases to help with extracorporeal membrane oxygenation (ECMO) therapy when patients become extremely hypoxic or their lungs are no longer functioning. Other types of hemodynamic support, including Impella and intra-aortic balloon pumps (IABP), also have been used to support COVID patients in cariogenic shock, in cases of left or right COVID-induced heart failure or multi-organ failure.
On April 6, 2020, the FDA issued guidance policy to expand the availability of ECMO to address the coronavirus emergency. This stated ECMO systems could be used for long-term respiratory/cardiopulmonary failure and provide assisted extracorporeal circulation and physiologic gas exchange of the patient’s blood for more than six hours. The technology can oxygenate a severely sick COVID-19 pneumonia patient's blood without the need to transfer the oxygen through fluid filled lungs. As a sudden acute respiratory syndrome, the FDA said COVID-19 can trigger acute respiratory failure and/or acute cardiopulmonary failure.
The FDA cleared LivaNova's ECMO therapy for longer than six hour in COVID patients April 23.
The Abiomed Impella RP has also been used to treat some COVID-19 patients suffering right heart failure. Acute PE may lead to acute right ventricular failure.
Fried said COVID-19 infection can cause decompensation of underlying heart failure, which might and lead to mixed shock. So the suggestion from the Columbia experience with COVID is to use invasive hemodynamic monitoring, if feasible, to better manage the cardiac component of shock in those patients.[3]
Older Critically Ill COVID-19 Patients May Have Increased Risk of Bradycardia With Lopinavir and Ritonavir
(UPDATED July 15, 2020) Older, critically ill patients with COVID-19 who received a combination of the antiretroviral medications lopinavir and ritonavir were more often seen to experience bradycardia, a slow heart rate, according to new research published today in Circulation: Arrhythmia and Electrophysiology, an American Heart Association (AHA) journal.[7]
The combination of antiretroviral medications lopinavir (LPV) and ritonavir (RTV) have been previously used to treat patients with SARS-Cov-1 and MERS-Cov, as well for HIV-1 patients. Among HIV-1 patients, a risk of bradycardia was also reported, researchers said.
In this small, preliminary, prospective study, researchers recorded the risk of bradycardia in critically ill COVID-19 patients treated with this combination of medications. Bradycardia is classified as a heart rate below 60 beats per minute for a period of more than 24 hours. Bradycardia can cause problems if the slow heart rate leads to a decrease in blood flow to the body. This can lead to heart failure, fainting, chest pain and low blood pressure. In some people, bradycardia does not cause any symptoms.
The study included 41 patients with COVID-19 admitted to the intensive care unit at Amiens University Hospital, in Amiens, France, who were treated with 200 mg LPV and 50 mg RTV twice daily for 10 days. All patients received continuous electrocardiogram (ECG) monitoring.
Among the patients who received the LPV/RTV treatment:
• 22% experienced bradycardia for more than 24 hours.
• Bradycardia occurred at least 48 hours after initiation of treatment, indicating that the medications may have caused bradycardia.
• Blood test measuring the concentration of ritonavir (RTV plasma concentration) at 72 hours after receiving the treatment showed higher concentrations of RTV in the patients who had bradycardia.
• No correlation was found between RTV plasma concentration, LPV plasma concentration and mean heart rate at 3-days after LPV/RTV treatment began.
• Patients experiencing bradycardia were on average older than patients receiving the treatment who did not experience bradycardia (ages 62-80 vs. 54-68, respectively).
• Bradycardia resolved after LPV/RTV were discontinued or doses were reduced.
“[LPV and RTV] have complex pharmacokinetic characteristics [how the body processes a medication]... Bradycardia could be a sign of severe cardiological or neurological impairment since it is associated with lymphopenia [lower than normal number of white blood cells] that seems to reflect the severity of COVID-19 infection. Intensivists should be aware of this potential side effect in order to closely monitor LPV/RTV plasma levels, notably in elderly patients," researchers noted in the study.
COVID Can Cause Both Ischemic and Hemorrhagic Strokes
There have been several studies published on COVID-19 patients presenting with stroke. In the largest study to date from Italy that included 725 consecutive hospitalized COVID-19 patients, 108 (15%) had acute neurological symptoms requiring neuroimaging.[6] The most common neurological symptoms were altered mental status in 64 (59%) patients and ischemic stroke in 34 (31%) patients.
The study also found six patients (6%) who had intracranial hemorrhage, and two had cerebral venous thrombosis.
Read more in the article How COVID-19 Effects the Brain in Neuroimaging
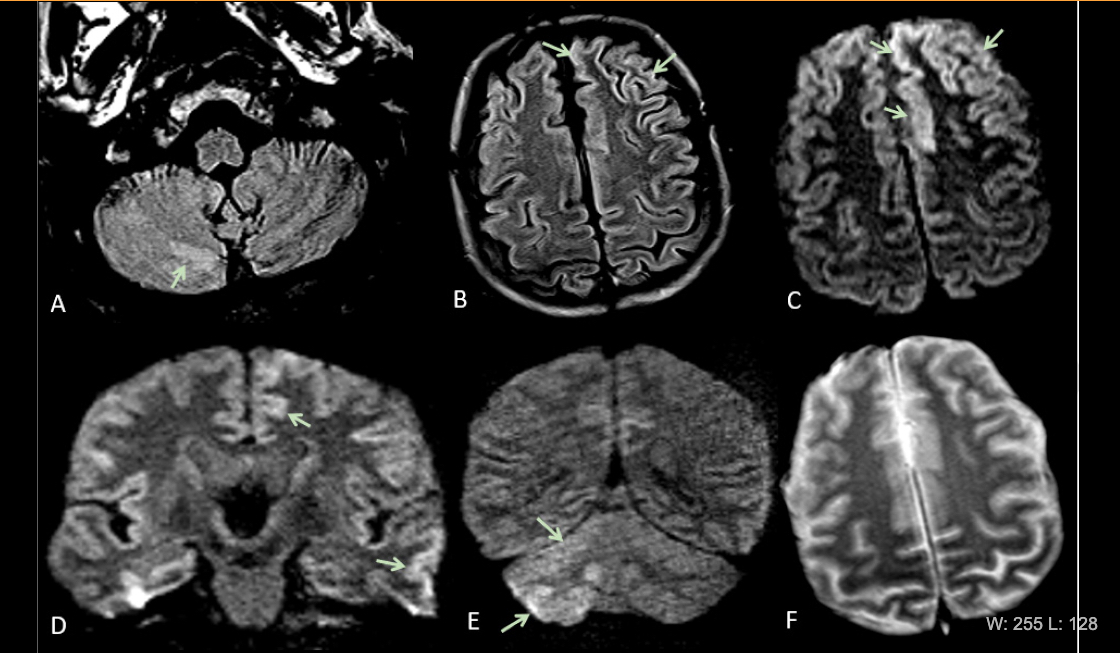
Images showing the cerebral vascular impact of COVID-19. Acute encephalopathy in a 60 year-old-man without history of seizures presenting with convulsion. (A-B) Multifocal areas of FLAIR hyperintensity in the right cerebellum (arrows in A), left anterior cingular cortex and superior frontal gyrus (arrows in B). (C-D) Restricted diffusion in the left anterior cingulate cortex, superior frontal and middle temporal gyrus (arrows in D) and right cerebellum (arrows in E), consistent with cerebellar diaschisis. F) No hemosiderin deposits in gradient echo sequences. Image courtesy of RSNA journal Radiology.
COVID-19 Causes Heart Attack Patients to Stay at Home
Patients' fear of catching COVID also has greatly impacted the usual STEMI volumes at hospitals. This sudden drop of STEMI cases by 40-60 percent was observed in Italy, Spain and the U.S. in April and May, causin g great concern by medical societies that this will cause a large number of unnecessary deaths, or outcomes of patients who survive will be very poor.
g great concern by medical societies that this will cause a large number of unnecessary deaths, or outcomes of patients who survive will be very poor.
Hospitals nationwide are seeing up to a 60 percent reduction in admissions for heart attacks, according to the American College of Emergency Physicians. Other emergency room volumes are also down by up to 50 percent, underscoring fears in the medical community of the dangerous, and potentially fatal, effects of COVID-19. These fears are also backed by a recent study showing a 38 percent drop in patients being treated with a life-threatening ST-elevated myocardial infarction (STEMI) heart attacks.
"Regular STEMI acute myocardial infarction have disappeared," O'Neill said. "It is really bizarre, who knows where they are. Some people are just too afraid of coming down to the hospital."
A national SCAI survey of the public in May found as states begin to re-open and the number of COVID-19 cases declines, 40 percent of Americans still view going to the hospital as a risky behavior — even more than going to the hair salon, a concert or the beach. SCAI and the ACC both launched public service campaigns encouraging heart attack and stroke patients call 911 go to the hospital and not to worry about COVID because precautions are taken to minimize infection.
“Cardiovascular disease is not hiding out in self-isolation waiting until it’s safe to strike,” said Kirk N. Garratt, M.D., MSc, MSCAI, medical director, Center for Heart and Vascular Health, Christiana Care Health System in Newark, Del., and SCAI past president. “Now more than ever, we need to make sure people at risk, and their loved ones, know the signs of a heart attack or stroke and understand the need to get to the hospital quickly. Fear of COVID-19 can also be fatal. Delaying care results in more serious heart damage and even death. Our hospitals are ready to give the right cardiac care – safely.”
Read more on the missing STEMIs
Watch the VIDEO: Where Have all the STEMI Cases Gone Amid COVID-19? — Interview with Thomas Maddox, M.D.
Related COVID-19 Cardiology Content:
How Cardiology Dealt With the COVID-19 Surge in New York City
Kawasaki-like Inflammatory Disease Affects Children With COVID-19
New Research Highlights Blood Clot Dangers of COVID-19
Cardiology Stands Up to the COVID-19 Challenge
VIDEO: Impact of COVID-19 on the Interventional Cardiology Program at Henry Ford Hospital — Interview with William O'Neill, M.D.
ACC COVID-19 Clinical Guidance For the Cardiovascular Care Team
The Cardiac Implications of Novel Coronavirus
Impella RP Granted FDA Emergency Use Authorization for COVID-19 Patients With Right Heart Failure
COVID-19 STEMI Registry Created to Study Acute Cardiovascular Effects of the Virus
FDA Reports of Deaths and Injuries From Use of Antimalarial hydroxychloroquine in COVID-19 Patients
How to Manage AMI Patients During the COVID-19 Pandemic
References:


 March 04, 2026
March 04, 2026 









