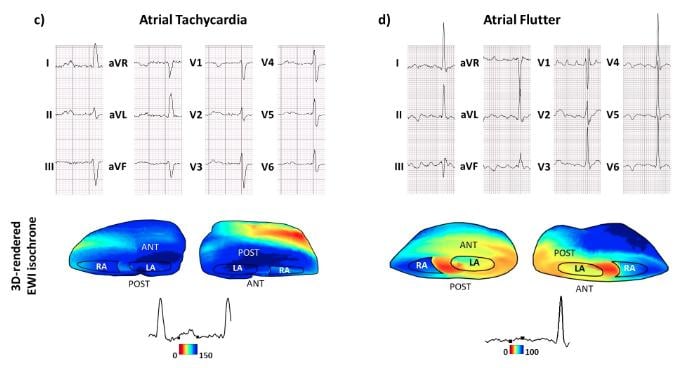
Electromechanical Wave Imaging (EWI) is a new, high-frame rate 3-D rendered ultrasound technique that can noninvasively map the electromechanical activation of heart rhythm. This example shows the ECG tracings compared to the EWI image of the heart.
May 15, 2019 — A new study shows electromechanical wave imaging is capable of localizing arrhythmias including atrial tachycardia (AT), atrial flutter (AFL), premature ventricular complexes (PVC), and accessory pathways (AP) in Wolff-Parkinson-White syndrome prior to catheter ablation. The double-blinded study for this new technology was presented as a late-breaking session at Heart Rhythm 2019, the Heart Rhythm Society's 40th Annual Scientific Sessions.
Diagnosis and localization of arrhythmias are critical for decision making and treatment planning prior to cardiac ablation. The 12 lead ECG is the main noninvasive tool used to localize arrhythmias, however it can be limited in specificity and subject to interobserver variability. Electromechanical Wave Imaging (EWI) is a novel, high frame rate 3-D rendered ultrasound technique that can noninvasively map the electromechanical activation of heart rhythm.
In this study, EWI was able to correctly predict 96% of arrhythmia locations as compared with 71% for 12-lead ECG analyses (unadjusted for arrhythmia type: OR: 11.8; 95% CI: 2.2-63.2; p=0.004; adjusted for arrhythmia type: OR: 12.1; 95% CI: 2.3-63.2; p=0.003). There was significant inter-observer variability amongst ECG reads.
Ongoing efforts include incorporating EWI isochrones into 3-D anatomical maps prior to ablation to determine whether EWI can affect clinical outcomes, said Elaine Wan, M.D., FHRS, the Esther Aboodi Assistant Professor of Medicine in Cardiology and Cardiac Electrophysiology at Columbia University Medical Center. She explained this includes reducing procedure time and radiation dose, in addition to exploring EWI with transesophageal, intracardiac, and 3-D ultrasound.
In the study, patients presenting for catheter ablation of AT, AFL, PVCs, or an AP underwent transthoracic EWI and 12 lead ECG prior to the ablation. 67 patients were consented, 12 patients were excluded due to withdrawal of consent, poor imaging or not being in the arrhythmia at time of imaging. There were 55 patients who underwent EWI (mean age 56.0±2.3 years, 71% male). Of these, 22% had WPW, 20% had PVCs, 13% had AT and 45% had AFL. Six blinded clinical electrophysiologists predicted arrhythmia origins based on ECG alone. EWI isochrone processing was blinded to all clinical data except arrhythmia type. Operating physicians were blinded to EWI isochrones. EWI and ECG predictions were compared to arrhythmia location as determined by 3-D electroanatomic mapping and ablation using a standardized 21 segment cardiac model.
All the HRS 2019 late-breaking studies



 May 06, 2026
May 06, 2026 









