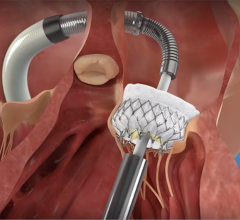
Structural heart interventions represent one of the fastest growing segments of cardiology, as the number of devices to address valvular and nonvalvular heart disease continues to expand rapidly. Imaging is a critical component for all aspects of structural heart treatment — from disease assessment and patient selection, to device selection and sizing as well as procedural risk prediction.
The Growth of CT in TAVR
Minimally invasive or transcatheter interventions offer numerous benefits for patients who are not suited for cardiovascular surgery, including quicker recovery time in many cases. Unlike with surgery, transcatheter approaches do not allow the interventionist to see the area they are treating, so they must rely on imaging to plan and guide the procedure.
Transcatheter aortic valve replacement (TAVR) has largely been the driving force behind the growth of structural heart interventions (and structural heart imaging) in recent years. Once just an option for those unsuitable for surgery, various clinical studies have expanded the patient population for TAVR, down to intermediate- and even some low-risk patient cohorts.
The importance of computed tomography (CT) has also grown as TAVR continues to expand. Structural heart imaging is crucial when answering several key questions as a procedure is being planned, including:
• Is the patient a good candidate for the procedure?
• Does the patient have the appropriate landing zone for the device in question?
• Are they at risk for procedural complications?
• How big of a device is needed to complete the procedure?
Angiography has traditionally been used in interventional scenarios because it offers real-time visualization. The drawback of fluoroscopy is that it offers little in the way of soft tissue definition. This is largely why CT has become the gold standard for answering the above questions when planning TAVR and other structural heart interventions.
The Society of Cardiovascular Computed Tomography (SCCT) released an updated consensus document in January 2019 as a guide to using CT to improve patient outcomes during TAVR. According to SCCT, the document is meant to reflect the wealth of data published since the last update in 2012 on CT and TAVR. The 2019 document contains recommendations on:
• CT acquisition prior to TAVR;
• Sizing and reporting of the aortic valve, annulus and outflow tract;
• Reporting of fluoroscopic angulation;
• Reporting of vascular access, coronary artery and non-cardiac, non-vascular findings; and
• Post-TAVR pre-VIV (valve-in-valve) scans.
“The role of computed tomography, which was initially used primarily for evaluating peripheral access, has grown substantially,” said author Philipp Blanke, M.D., FSCCT, of the University of British Columbia. “It is now the gold standard for accurately sizing the annulus, determining the risk of annular injury, and coronary occlusion and providing predictions in advance of the TAVI/TAVR procedure.”
Expanding Into Mitral and Tricuspid Valves
The maturity of TAVR technology and the understanding of the aortic space has allowed focus to shift toward transcatheter procedures in the mitral and tricuspid spaces, which pose unique challenges of their own.
“I’ve had the opportunity to learn, especially in the last decade with the integration of CT in [TAVR], and now we’re applying those lessons to the transcatheter mitral and tricuspid space,” said
Jonathon Leipsic, M.D., FSCCT, chairman of the department of radiology, St. Paul’s Hospital, Vancouver, Canada.
Transcatheter mitral valve interventions (TMVI) have seen a great deal of innovation and study in recent years, with several devices receiving U.S. Food and Drug Administration (FDA) approval since the first TMVI in the U.S. in 2015. The anatomy of the mitral valve offers several complications for transcatheter replacement, such as a saddle-shaped annulus, integration into the left ventricle with chordal attachments and other challenges. “CT really provides an entire roadmap for all of those anatomical features, and also the perivalvular structures,” said Leipsic.
LVOT Obstruction From Mitral Implants
One major consideration for the mitral space not present in the aortic space is left ventricular outflow tract (LVOT) obstruction, a potentially lethal procedural complication. CT can be used in pre-procedural planning to calculate the neo-LVOT, or the residual area that remains after valve implantation. According to Leipsic, several factors come into play when calculating the neo-LVOT, including:
• How far the device extends into the ventricle;
• Whether the device tilts toward or away from the LVOT during deployment;
• Volume status;
• Septal thickness; and
• Residual ejection fraction (EF)
“But over the years what we realized is that we didn’t want to just provide a diameter, but rather an assessment of the overall residual area following a virtual implantation,” said Leipsic.
Use of TEE in Mitral Repairs
While CT plays a role in the mitral space, transesophageal echocardiography (TEE) has emerged as the key modality for peri-procedural guidance. This is due to the proximity of a TEE probe to the left atrium, which provides an unobstructed view of the procedural area.
The necessity of TEE is bringing echocardiographers into the cath lab and may require a shift in thinking and procedures. “I think this is the advent of a new subspecialty for us and I think we are still in the development phase of what is needed for the qualifications of the individual that is going to participate in these procedures,” said
Rebecca Hahn, M.D., FASE, Columbia University Medical Center, New York, and a key thought leader on interventional echo.
Structural Heart Imaging Software Considerations
While the imaging modalities themselves are important for structural heart assessment, equally so is the software being used to mine the data and create treatment plans. With a wide variety of software options available, hospitals — especially those just starting a structural heart program — should find something that best suits their specific needs and workflows.
“I think what people should look for is a software that’s user-friendly, that has a very straightforward workflow and that yields very robust reproducibility,” said Leipsic. He offered a few examples of what to look for when choosing structural heart planning software:
• The software will be used for device sizing and selection as well as anatomical measurement, so it must be able to represent these numbers accordingly. Any software should be able to assess things like the size of the valve annulus and coronary ostial height (to understand the risk of coronary occlusion). “Most of the workstation algorithms can provide solutions for this,” said Leipsic.
• He added that workflow and reproducibility go hand in hand, as accurate results will streamline diagnoses and, in turn, patient care. Imaging software can help achieve this through features such as a smoothing algorithm for perimeter to measure the aortic annulus, with the ability to generate S-shaped curves of perpendicularity or coplanarity.
• In the mitral space, with its more complex anatomy, reproducibility is even more important. Software should be able to offer reproducible segmentation of the mitral annulus as well as the neo-LVOT to predict the risk of LVOT obstruction.
• Many softwares now allow users to test a device’s suitability through virtual sizing and placement. This can help boost procedural confidence and ensure the proper device is placed correctly the first time.
Augmented Reality Might Be The Next Frontier
While 3-D and 4-D imaging offer improved detail for structural heart cases, it is still not the same thing as being able to physically see the target area as in surgery. The next generation of imaging systems are exploring the use of augmented reality (AR) to address these shortcomings.
Augmented reality superimposes a virtual 3-D image on top of the physical environment, typically through a headset or other smart device. The technology is being developed for use in numerous industries, and healthcare applications are focused on procedural guidance. As it has been displayed, the idea is that the surgeon or interventionist can see imaging data from CT, MRI or other modalities displayed directly on top of the patient holographically.
The most recent display of AR healthcare technology came from Philips and Microsoft at Mobile World Congress in Barcelona, Spain, in February. Microsoft’s HoloLens 2 mixed reality platform — a self-contained holographic computer that allows hands-free, heads-up interaction with 3-D digital objects — was used in tandem with Philips’ Azurion image-guided therapy platform to allow physical interaction with live imaging and other data.
TeraRecon launched its augmented reality system, the HoloPack Portal, at the 2017 Healthcare Information and Management Systems Society (HIMSS) annual meeting. The system, which also uses the Microsoft HoloLens visor, extends TeraRecon’s existing 3-D viewing capabilities to real 3-D image viewing in the visor. The system uses voice controls and finger movements in the air to manipulate 3-D holographic objects. This could potentially allow clinicians to manipulate anatomical images without breaking the sterile field, and then glance down under the visor to continue the procedure.
The first HoloLens-based augmented reality system to receive FDA clearance was Novarad’s OpenSight, cleared last October. The system was approved for pre-operative surgical planning, rendering 2-D, 3-D and 4-D images that are co-localized onto the real-world patient. Multiple headsets can be shared among users to provide enhanced procedural training.




 July 08, 2024
July 08, 2024