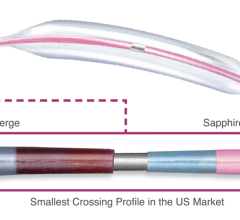
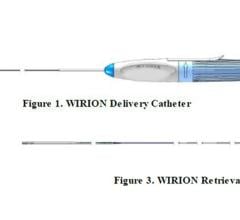
The Intact Vascular Tack Endovascular System is designed to spot stent dissections with a short, self-expanding 6 mm long stent that can expand to vessel diameters between 2.5-6 mm, so exact sizing is not needed.
It is estimated that more than 10 million people in the United States are affected by peripheral arterial disease (PAD),[1] and these patients represent a new source to boost cath lab volumes. However, very high rates of restenosis in PAD lesions have necessitated the development of new tools to improve vessel patency rates.
PAD blockages can result in severe pain, limited physical mobility and non-healing leg ulcers, and lead to amputation if the vessels cannot be revascularized. More than 1 million Americans are living with a limb loss as a result of diabetes and PAD,[2] and nearly half of these patients die within five years.[3] About 150,000 Americans undergo leg or foot amputations every year, mostly due to poor circulation caused by PAD.
About 25 percent of PAD cases will progress to critical limb ischemia (CLI), which is associated with complex occlusions in the tibial arteries. It is estimated that up to 20 percent of these occlusions are unable to be treated from the traditional femoral artery access site. However, recent advancements in technology and techniques have made the use of retrograde tibiopedal access (where physicians achieve access through the arteries in the foot or ankle) more popular, thereby expanding the treatment options available for PAD patients.
Watch the video, “State of Therapy for Critical Limb Ischemia.” This interview with Michael Jaff, M.D., medical director of Massachusetts General Hospital’s Fireman Vascular Center, explains the current trends and technologies in critical limb (CLI) ischemia at ACC.16. Jaff also discusses strategies used by hospitals to recruit patients into CLI programs.
“In recent years there has been a revolution in endovascular therapy and we’re able to treat calcified and smaller vessels, meaning we can significantly improve blood flow and save limbs that once weren’t considered salvageable, and that saves lives,” said Julio Sanguily III, M.D., vascular surgeon with Martin Health System, Stuart, Fla., who presented a study on CLI at the 2015 International Symposium on Endovascular Therapy (ISET).
He explained that a good percentage of these patients not only have limbs salvaged but their quality of life greatly improved through interventional techniques. Sanguily said the key is to educate physicians who treat elderly patients and those with diabetes or kidney failure, the patients at highest risk for these problems. He said letting them know about new interventional options and providing them the ability to refer patients to a limb salvage program can help rapidly build a program. The number of amputations at Martin Health System fell by 79 percent over five years after the center implemented a limb salvage program. In 2014, he said 500 patients were evaluated with an angiogram to assess blood flow in the legs. Of these patients, only five had amputations (1 percent).
Whenever possible, PAD patients in the Martin Health System program received treatment to revascularize the legs, including percutaneous transluminal angioplasty (PTA), atherectomy and hyperbaric oxygen therapy. Sanguily said calcified plaque is the most challenging problem encountered in these interventions and atherectomy has proved particularly helpful in these lesions.
Here are some of the recent advances in PAD treatment technology.
Peripheral Stents
The revascularization strategy for PAD differs from coronary artery disease (CAD). Instead of attempting a permanent repair to reopen vessels as in CAD, the vessels in the leg are treated with the expectation that the patient will need the same segments revascularized again in the future. PAD restenosis rates are very high with the use of both PTA and stents. Stents in the legs are exposed to torque and pressures not found in the heart, protected by the ribcage. This environment leads to high rates of in-stent restenosis and stent fractures.
These issues have led to development of specialty stents for PAD. Most peripheral stents are self-expanding, flexible and handle flexing and external crushing pressures better than rigid balloon expanded stents.
One of the most recent stents approved by the U.S. Food and Drug Administration (FDA) in December was Biotronik's Astron for iliac lesions. The self-expanding stent is loaded on an over-the-wire delivery system available in diameters of 7, 8, 9 and 10 mm, and stent lengths of 30, 40, 60 and 80 mm. Astron’s 12-month primary patency rate was 89.8 percent, and the target lesion revascularization (TLR) rate was 1.4 percent. Additionally, patients demonstrated improvement in their ambulatory function and quality of life at 12 months compared to baseline as evaluated by the walking impairment questionnaire score, a common PAD screening tool.
In February, Biotronik announced a partnership with Maquet Medical Systems in which Maquet will expand its product portfolio into PAD using Biotronik’s devices. In 2014, an estimated 176,800 patients in the United States received an iliac stent, and that number is expected to grow annually, offering new growth opportunities for companies with PAD portfolios.[4]
Last August, Boston Scientific received FDA clearance for the Innova self-expanding stent for the superficial femoral artery (SFA) or proximal popliteal artery (PPA). The platform consists of a nitinol self-expanding bare metal stent available in diameters from 5-8 mm and lengths of 20-200 mm. This stent platform serves as the foundation for the Eluvia drug-eluting vascular stent in development. The SFA and PPA present a challenging environment for stents, and the flexibility, radial strength and fracture resistance of the Innova Stent are designed specifically for this anatomy.
One-year clinical trial outcomes assessing the Eluvia drug-eluting stent reflect a primary patency rate of more than 96 percent. These results represent the highest 12-month primary patency reported for an interventional treatment of femoropopliteal artery lesions among comparable trials. Results from the MAJESTIC trial were presented at the 2015 Cardiovascular and Interventional Radiological Society of Europe (CIRSE) meeting last fall. Data included a low 12-month TLR rate of 3.8 percent, with no observed stent fractures and no amputations. The prospective, multicenter clinical trial enrolled 57 patients across Europe, Australia and New Zealand, with an average lesion length of 70.8 mm. The trial included a high percentage of complex lesions, with 46 percent of lesions classified as total occlusions and 65 percent identified as severely calcified. Data from the trial are expected to support global regulatory submissions.
Boston Scientific has received an investigational device exemption (IDE) from the FDA to begin a global, pivotal study of the Eluvia drug-eluting stent. The IMPERIAL trial, designed to support future regulatory submissions and patient enrollment, is expected to begin in the coming months.
The FDA cleared Covidien’s EverFlex self-expanding stent in 2014. The device is indicated for use in the SFA and PPA. It is available in a size matrix from 20-200 mm and is deliverable through a 6 French catheter. Its spiral-cell connection and peak-to-peak connection nodes are designed to disperse force uniformly, while the three-wave peak design resists compression and provides good wall apposition. Medtronic has since acquired Covidien, adding the stent and the rest of Covidien’s PAD products to its portfolio.
Medtronic also received FDA clearance for the Protege GPS self-expanding stent in early 2015. The stent is indicated for lesions of the common and external iliac arteries. The news followed the nine-month results of the DURABILITY Iliac study, presented at the 2014 VIVA (Vascular Interventional Advances) meeting. The prospective, multi-center, non-randomized clinical study demonstrated 95.8 percent nine-month primary patency by Kaplan-Meier analysis and 98.6 percent freedom from target vessel revascularization.
The first drug-eluting, self-expanding stent, approved by the FDA in 2012, was the Cook Medical Zilver PTX. Five-year data from the Zilver PTX Randomized Controlled Trial of Paclitaxel-Eluting Stents for Femoropopliteal Disease showed primary patency of 66.4 percent in the SFA. This compares to 43.4 percent patency for patients with PTA or provisional bare metal stent placement. The multicenter, prospective, randomized study included 479 patients.
Drug-coated Balloons
The FDA approved the first two drug-coated balloons (DCB) for the U.S. market in late 2014. C.R. Bard’s Lutonix and Medtronic’s IN.PACT Admiral DCBs are both indicated to treat the SFA. Since peripheral stents traditionally have high restenosis rates, DCBs offer the ability to treat a vessel segment and deliver the antiproliferative drug paclitaxel to the vessel wall without the use of a stent. Long-term DCB clinical data continues to mount, showing DCBs have superior outcomes over conventional PTA.
The IN.PACT SFA study was presented as a late-breaking trial at the 2015 Transcatheter Cardiovascular Therapeutics (TCT) symposium last fall. Its two-year results showed DEBs were superior to traditional PTA. The prospective, multicenter, single-blinded trial enrolled 331 patients in a 2:1 randomization to a DCB group or standard PTA. The primary efficacy endpoint was primary patency, defined as freedom from TLR and duplex-derived restenosis. At two years, patients treated with DCB showed higher primary patency compared to PTA (78.9 vs. 50.1 percent). Freedom from TLR was 91 percent for DCB compared to 72.2 percent for PTA. Results of the two-year primary safety composite were also positive for the DCB group (87.4 vs. 69.8 percent). There were no device- or procedure-related deaths and no major amputations in either group through two-year follow-up, although mortality unrelated to the device/procedure was higher with DCB compared with PTA alone. In addition, DCB patients achieved similar functional outcomes with 58 percent fewer re-interventions.
“The two-year data from IN.PACT SFA demonstrates the continued superiority of drug-coated balloons for treating patients with superficial femoral artery disease compared to percutaneous transluminal angioplasty,” said lead investigator John R. Laird Jr., M.D., professor of medicine and medical director of the Vascular Center at UC Davis Medical Center. “With a sustained durability and no late catch-up after two years, this has the potential to drive a paradigm shift in how we treat SFA disease.”
Results from the LEVANT 2 trial, published in the June 2015 online edition of The New England Journal of Medicine, demonstrated superior primary patency for the Lutonix DCB over standard PTA, as well as safety consistent with standard PTA balloons. This global, prospective, single-blind, randomized, 54-site study (42 sites in the United States and 12 in Europe) enrolled all patients under one protocol, comparing the Lutonix 035 DCB with standard PTA. The study demonstrated sustained improvements in Rutherford category from baseline to 12 months, and improved patient-reported walking distance scores.
One obstacle to broad use of DCBs in the United States was the initial lack of reimbursement. However, the Centers for Medicare and Medicaid Services (CMS) approved a transitional pass-through payment for both the Lutonix and IN.PACT Admiral devices in 2015. The purpose of the reimbursement is to cover additional cost to U.S. hospitals for treating Medicare beneficiaries with the Lutonix DCB in the outpatient setting. CMS determined that costs associated with DCBs were not included in existing reimbursement for PTA, stenting or atherectomy procedures. Therefore, CMS removed the device offset charge from the calculation and now reimburses the full cost of DCBs in these procedures.
Last spring, Spectranetics Corp. accelerated investments totaling about $6 million in its Stellarex drug-coated balloon angioplasty (DCB) platform for treatment of below the knee (BTK) disease. The company estimates BTK disease will represent a $150 million market opportunity by 2020, and the company anticipates the global market for DCBs to reach between $700 million and $1 billion over the next seven years. Spectranetics is targeting European approval for the Stellarex DCB BTK platform in the second half of 2016. The company expects to commence enrollment in a U.S. IDE clinical trial in mid-2016.
The Stellarex uses EnduraCoat technology, a durable, uniform coating designed to prevent drug loss during transit and facilitate controlled, efficient drug delivery to the treatment site.
Atherectomy Systems
Two new atherectomy systems were approved in the past couple years, offering additional treatment options to debulk peripheral lesions with or without stenting, especially calcified lesions.
In April 2015, the FDA cleared the Cardiovascular Systems Inc. (CSI) 4 French, 1.25 Solid Diamondback 360 Peripheral Orbital Atherectomy System (OAS). Specifically, the clearance covers the company’s 145-cm-long, 1.25 mm Diamondback 360 Solid Crown, 4 French sheath compatible and the modified 1.25 mm Diamondback 360 Micro Crown, 4 French sheath compatible systems. Both devices offer a smaller profile and a more flexible shaft for improved ease of use. The use of smaller access sheaths can help reduce procedure times, enable quicker patient recovery and result in fewer procedural complications from bleeding.
Last July, the FDA cleared CSI’s ViperWire Advance Peripheral Guide Wire with Flex Tip for the Diamondback. The new guide wire provides improved flexibility, navigation and ease-of-use, particularly in hard-to-reach, tortuous vessels when treating arterial calcium.
The FDA cleared Covidien’s HawkOne directional atherectomy system and it was released commercially in April 2015. The HawkOne provides an enhanced cutting mechanism to more effectively treat the widest variety of PAD plaque. The Covidien atherectomy portfolio includes the TurboHawk and SilverHawk systems.
Recent clinical studies have shown atherectomy can be safe and effective as a frontline PAD therapy. Results from the DEFINITIVE LE study using the TurboHawk and/or SilverHawk systems demonstrated 95 percent limb salvage in patients with CLI, and 78 percent overall patency in claudicant patients at 12 months. The DEFINITIVE LE study is the largest atherectomy study conducted to date with independent, core lab analysis of the clinical outcomes.
“The DEFINITIVE LE study provides confirmatory evidence based on 12-month, independent core lab adjudicated data on the use of directional atherectomy in treating a diverse, real-world group of patients with PAD,” said James F. McKinsey, M.D., FACS, co-global principal investigator and lead author of the study. “The study enrolled 800 patients, making it the largest trial with independent, core lab analysis for atherectomy to date. Results of the study demonstrate high limb salvage, patency rates and diabetic outcomes. This further confirms the effectiveness and versatility of directional atherectomy in a wide range of vessel sizes and clinical presentations. Atherectomy is an important treatment option as the therapy leaves nothing behind in the body and preserves all future treatment options.”
The prospective, multi-center DEFINITIVE LE study enrolled 800 patients in 47 centers in the United States and Europe and included two patient cohorts: those with claudication and those with CLI. For the first time in an atherectomy study, DEFINITIVE LE also included pre-specified sub-group analysis comparing patency rates in diabetic and non-diabetic patients.
“The outcomes for patients with claudication reveal that in the scientific landscape, directional atherectomy is not only safe but effective to a similar degree as previous trials in the superficial femoral artery (SFA). Diabetic patients, who remain challenging for therapy due to their advanced and multi-level disease, have similar patency rates to non-diabetic patients with claudication when performing directional atherectomy,” said Lawrence Garcia, co-global principal investigator of the study and chief of interventional cardiology and vascular interventions at St. Elizabeth’s Medical Center in Boston. “These findings provide strong clinical support for the versatility of directional atherectomy in treating patients with diabetes, claudication and critical limb ischemia.”
Among patients with claudication, primary patency was 78 percent at 12 months. Notably, this rate did not differ between patients with diabetes (77 percent) and those without diabetes (78 percent) — the first such results to be shown in a prospective, powered analysis. Specifically for the SFA, the patency rates were 83 percent in lesions under 10 cm in the claudicant cohort.
CSI announced in November 2015 that the first patients were treated in its OPTIMIZE study, a European study evaluating the acute and long-term clinical outcomes of orbital atherectomy with adjunctive DCB angioplasty versus DCB angioplasty. Specifically, the study will look at patients with calcified BTK lesions. OPTIMIZE also will compare the economic outcomes between the patient groups.
Novel Technologies to Treat Preipheral Artery Disease
Ballooning lesions can be very difficult when faced with calcified vessels. Overinflation to attempt breaking the calcium frequently results in vessel dissections. Two recent technologies may offer new PAD treatment options.
Intact Vascular was granted FDA permission to begin its IDE trial of the Tack Endovascular System. It is specifically designed for dissection repair in peripheral vessels. The Tack Optimized Balloon Angioplasty II (TOBA II) trial began treating patients last fall. It will enroll 210 patients at 30-40 sites in the United States and Europe.
The Tack Endovascular System (this system was cleared by the FDA in April 2019) spot-treats dissections with a short, 6 mm length self-expanding stent. These can be placed where needed to support the vessel and reduce the amount of metal left behind. This helps reduce the amount of inflammation and lowers the force on the vessel wall to reduce vessel trauma and reaction. The stents are also very flexible and will expand to fit arteries ranging from 2.5 to 6 mm in diameter, eliminating the need to precisely size the device, as is necessary with conventional stents.
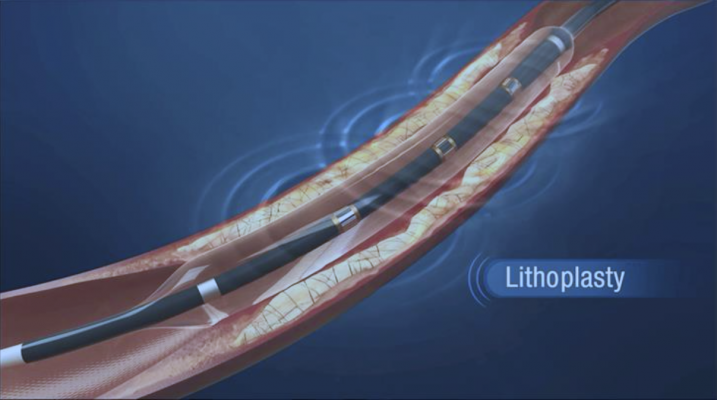
Another innovation to watch is Shockwave Medical’s Lithoplasty balloon catheters that use lithotripsy pulsatile mechanical energy bursts to shatter the calcium, rather than radial force of the balloon. Lithotripsy is commonly used to break up kidney stones and this system miniaturizes the energy delivery system to fit inside blood vessels. The balloon inflates to ensure good contact with the vessel wall, but not for dilatation. The shockwave pulse emitters inside the balloon’s core then activate to crack the calcium without additional balloon pressure. The technology is being developed for peripheral and coronary vessels, and in aortic valvuloplasty applications. Shockwave said it received $40 million in funding in 2015 to develop the technology.
The company reported six-month follow-up results in 2015 from its DISRUPT PAD trial, a study of 35 patients with calcified SFA and PPA stenosis treated with the Lithoplasty system. Data demonstrated safe and effective dilatation of calcified stenosis with no acute failures, favorable residual stenosis, no major device-related adverse events and no need for stent placement. Six-month durability was excellent, with no need for re-treatment and patency by duplex ultrasound of 83 percent.
Watch the VIDEO “Breaking Up Calcified Lesions Without Vessel Trauma.” A discussion with Todd Brinton, M.D., at TCT 2016 about the newly FDA-cleared Shockwave Medical Lithoplasty System. Brinton is clinical associate professor and adjunct associate professor of bioengineering at Stanford University Medical Center.
Shockwave received European approval for the Lithoplasty system in January 2015.
Support and CTO Catheters
Cordis launched its new Elitecross support catheter and Outback Elite re-entry catheter in the United States, Europe and Japan last summer. This expands the Cordis dedicated crossing portfolio for treatment of chronic total occlusions (CTO), a suite of specialty and workhorse solutions designed to support crossing complex lesions. The Elitecross features a braided shaft, tapered tip, lubricious hydrophilic coating and ultra low-friction inner lumen to enhance trackability over the guide wire. The device is designed to provide additional support to the distal portion of diagnostic or interventional devices, and is compatible with the Frontrunner XP CTO catheter as well as other ancillary devices.
The redesigned Outback Elite re-entry catheter enables faster and more precise re-entry into the true lumen in the most challenging cases. The device was redesigned with an ergonomic handle and torque control location, enabling single-handed operation by the user. The addition of an 80 cm shaft length will aid in optimizing procedures by reducing the length of shaft outside the patient. These new features combined with a redesigned package make the Outback Elite a more convenient, precision re-entry tool for tough lesions.
Cardinal Health acquired Johnson & Johnson’s Cordis business in 2015, helping expand Cardinal’s growing CAD and PAD portfolios
Reflow Medical received FDA clearance for its SpeX shapeable support catheter in December 2014. It enables physicians to shape the tip of the support catheter to their preferred angle to meet the specific needs of the case and maintain that angle throughout the procedure. It is designed to be compatible with physician-preferred 0.035 guidewires and the Wingman14 crossing catheters.
Roxwood Medical also received FDA clearance in December 2014 for its CenterCross Catheter for use in the coronary and peripheral vasculature. It is designed for use in conjunction with guidewires and microcatheters to access discrete regions of the coronary and peripheral vasculature and enable clinicians to better address wire-crossing of complex lesions. The catheter incorporates a unique self-expanding stent-like scaffold to stabilize off-the-shelf interventional tools, such as guidewires and microcatheters, in the center of the artery near the target lesion.
Roxwood reported in January that more than 500 patients have been successfully treated as part of the initial limited release of its MicroCross Catheter. It is offered in two sizes (Micro14 and Micro18) for use in the coronary and peripheral vasculatures, providing enhanced guidewire support through challenging and tortuous anatomy. Compatible with Rockwood’s CenterCross Catheter, MicroCross is comprised of an ultra-low profile shaft featuring variable pitch braid technology and a state-of-the-art lubricious coating. These features allow for enhanced trackability and guidewire support while navigating tortuous regions of the coronary and peripheral vasculature.
References:
1. "Peripheral Artery Disease." The Society for Cardiovascular Angiography and Interventions. http://www.scai.org/SecondsCount/Disease/PeripheralArteryDiseasePAD.aspx
4. "U.S. Markets for Peripheral Vascular Devices 2014." Millennium Research Group. 2013







 November 08, 2024
November 08, 2024