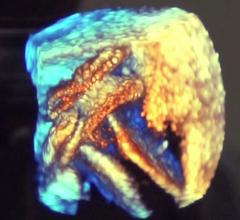
NMT Medicals BioStar bioabsorbable occluder, several months after implantation.
In addition to the FDA-cleared Gore Helex and the AGA Amplatzer transcatheter devices for atrial septal defects (ASDs), there are several others in development or already cleared for use outside the United States.
Produkte für die Medizin AG (PFM) currently offers the Nit-Occlud system in Europe. The spiral coil system is designed for transcatheter occlusion of patent ductus arteriosus (PDA) of all shapes. The company is now working on a patent foramen ovale (PFO) and ASD occluder, according to Ziyad Hijazi, M.D., MPH, professor of pediatric medicine, director of the Center for Congenital and Structural Heart Disease at Rush University Medical Center, Chicago. The company is also recruiting patients for a European trial using the Nit-Occlud to seal ventricular septal defects (VSDs).
NMT Medical’s CardioSeal and StarFlex are FDA-cleared to treat VSDs. The StarFlex is currently in U.S. trials to test its safety and efficacy in treating PFOs to prevent stroke/transient ischemic attack (TIA). The company also developed the bioabsorbable BioStar implant to seal atrial defects. It uses a set of self-centering microsprings and a collagen matrix. The collagen is absorbed by the body, only leaving behind the springs. The device is cleared for use in Canada and Europe.
The Coherex FlatStent has European approval to treat PFOs. The company has a distribution agreement with Abbott Vascular to distribute the device in Europe and Japan.
The Occlutech Figulla PFO and ASD occluder has clearance in Europe.
Hijazi said Gore is also working on a second-generation occluder device.





 June 20, 2024
June 20, 2024