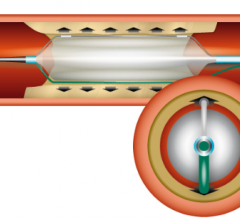
September 11, 2014 — Medtronic announced the U.S. Food and Drug Administration (FDA) 510(k) clearance and launch of the ...
Balloon Catheter
This channel includes news and new technology innovations for angioplasty balloon catheters (PTA). These are used in arteries with atherosclerotic lesions to compress the plaque expand the artery lumen to reopen occluded or heavily stenosed atherosclerotic lesions. Balloons are often used in combination with a stent to prop the treated vessel segment open. In addition to plain old balloon angioplasty (POBA), this section includes news about drug-coated balloon (DCB), valvuloplasty balloons and specialty cutting balloon.
August 12, 2014 — Teleflex Inc.’s subsidiary Hotspur Technologies Inc. received U.S. Food and Drug Administration (FDA) ...
July 28, 2014 — Boston Scientific Corp. received CE mark for the Ranger Paclitaxel-coated PTA Balloon Catheter. The ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
July 11, 2014 — Cordis Corp. announced the launch of its Saber PTA (percutaneous transluminal angioplasty) dilatation ...

June 16, 2014 — C. R. Bard Inc. announced that the U.S. Food and Drug Administration’s (FDA) Circulatory System Devices ...
June 3, 2014 — Medtronic Inc. announced CE mark and launch of the NC Euphora noncompliant balloon dilatation catheter ...
May 14, 2014 — OrbusNeich announced the launch of the Sapphire II NC coronary dilatation catheter, a non-compliant ballo ...
April 23, 2014 — Interim results from The Chocolate Bar Registry conducted in the United States demonstrate that use of ...
March 3, 2014 — Biotronik announced the release of its Passeo-18 Lux drug-releasing balloon (DRB) in all countries ...
February 28, 2014 — Cordis Corp. announced an agreement with TriReme Medical Inc. that grants the company exclusive ...


 September 11, 2014
September 11, 2014