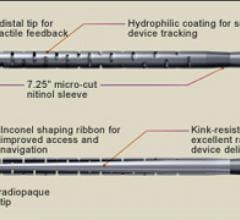
May 30, 2012 — MAQUET Cardiopulmonary, a business unit of MAQUET Cardiovascular and a leader in extracorporeal membrane ...
Catheters
January 20, 2012 - BridgePoint Medical Inc. this week signed an agreement with Covidien to exclusively license the ...
November 23, 2011 – Medical technology incubator Therix Medical announced the launch of spin-off Bluegrass Vascular ...
July 26, 2011 — Concentric Medical Inc. has launched the DAC 070 Catheter, the fourth and largest diameter addition to ...
May 26, 2011 – AngioDynamics announced launch of the new 90 cm NeverTouch procedure kit featuring a longer TRE-Sheath ...
May 19, 2011 - BridgePoint Medical Inc. has announced that they have received clearance of an expanded indication for ...
April 5, 2011 – A microcatheter system has received the CE mark. The Plato Microcath system, by Scientia Vascular ...
March 10, 2011 – A newly introduced catheter provides a superior answer for percutaneous fluid aspirations and ...
February 16, 2011 – To expand its portfolio of devices for lower extremity peripheral artery disease (PAD) ...
January 25, 2011 – A microcatheter offering superior crossability, flexibility and guidewire support during ...
January 13, 2011 – A catheter that removes emboli and thrombi from vessels in the coronary and peripheral ...
November 17, 2010 – A drug-eluting balloon (DEB) and a coronary stent have received CE mark approval in Europe ...
October 21, 2010 – Vascular Solutions has acquired the assets related to snare products from Radius Medical ...
October 13, 2010 – A new guidewire has been launched for use in challenging, small vessel peripheral angioplasty ...
October 5, 2010 – A U.S. clinical study evaluating the CrossBoss and Stingray catheters in treating chronically ...

 May 30, 2012
May 30, 2012