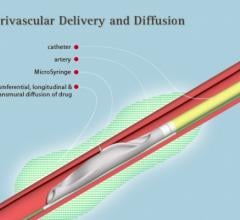
May 23, 2018 — Guerbet LLC USA announced the upcoming launch of SeQure and DraKon, two novel microcatheters for tumor ...
Catheters
There are few downsides to using tibial venous access to treat deep vein thrombosis (DVT) with catheter-directed ...
January 8, 2018 —Guerbet announced that it has entered into an agreement under which it will acquire Israeli company ...
January 4, 2018 — The U.S. Food and Drug Administration (FDA) announced a Class I recall of Sterilmed’s Agilis Steerable ...
As payers and other healthcare entities look to better manage costs, especially in the acute care setting, it is ...
November 2, 2017 — Crossing the occlusion with a guidewire is often the most challenging part of chronic total occlusion ...
October 5, 2017 — BTG plc announced it has acquired Roxwood Medical, provider of advanced cardiovascular specialty ...
October 4, 2017 — TVA Medical Inc. announced that its everlinQ 4 endoAVF System has received CE Mark in the European ...
October 2, 2017 — Reflow Medical Inc. announced that the company has received 510(k) clearance from the U.S. Food and ...
August 9, 2017 — Roxwood Medical Inc. recently announced it has entered into an exclusive agreement with Abbott for ...
June 28, 2017 — Royal Philips recently announced the relaunch of its Pioneer Plus catheter, the first and only re-entry ...
June 22, 2017 — The U.S. Food and Drug Administration (FDA) has identified Vascular Solutions’ recent recall of its ...
March 2, 2017 — Teleflex Inc. has announced 510(k) clearance by the U.S. Food and Drug Administration (FDA) and U.S ...
February 17, 2017 — Medtronic plc announced that its coronary portfolio will now include the DxTerity Diagnostic ...
February 15, 2017 — Mercator MedSystems announced that the national co-principal investigators of the company’s DANCE tr ...

 May 23, 2018
May 23, 2018