The H3+ digital recorder redefines “small” for Holter recorders while providing 2- and 3-channel recordings, an ...
EP Lab
This channel includes news and new technology innovations for cardiac electrophysiology (EP) systems, techniques and devices using in EP labs. This includes implantable EP devices, pacemakers, implantable cardioverter defibrillators (ICD), cardiac resychronization therapy (CRT), ablation technologies, left atrial appendage (LAA) occlusion, atrial fibrilation (AF) and Holter monitors.
Mortara’s H12+ digital Holter record provides 12-lear data recorded beat by beat on a compact flash memory card ...
February 25, 2009 - The HemCon Patch, a flexible hemostatic dressing from HemCon Medical Technologies Inc., is ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
February 16, 2009 - Registration is now open for the Heart Rhythm Society’s free Reality EP Webcast, “Role of 3D ...
February 12, 2009 - Sanofi-aventis said yesterday the ATHENA trial was published in the New England Journal of ...
February 12, 2009- Transoma Medical’s Sleuth AT (Advanced Trending) cardiac monitoring system, the company’s ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
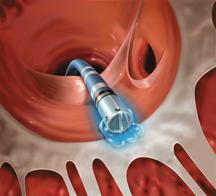
The practice of cardiac catheter ablation may experience a big boost as catheter ablation systems are poised to be ...
February 11, 2009 - Medtronic Inc.’s Reveal XT Insertable Cardiac Monitor (ICM) is now commercially available in ...
ST Cardio Technologies’ Z6 Electrophysiology Stimulator is an advanced Windows XP based, digitally controlled ...
Washington Health System (WHS) provides healthcare services at more than 40 offsite locations across three counties in ...

February 9, 2009 - Biosense Webster Inc. said Friday the FDA granted market clearance for the NaviStar ThermoCool ...
February 6, 2009 - Biosense Webster Inc., a Johnson & Johnson company, today said the FDA granted market approval ...
February 6, 2009 – The FDA has approved St. Jude Medical Inc.’s medical device system that allows a single ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
February 3, 2009 - New research proves increased QT variability is an independent predictor of ventricular ...
February 4, 2009 - On Thursday, Feb. 19 at 7:00 pm, The Halifax Health - Center for Cardiology, Daytona Beach, FL ...
January 29, 2009 - Medtronic Inc. today reported the expansion of its CURE-AF (Concomitant Utilization of ...

 February 26, 2009
February 26, 2009