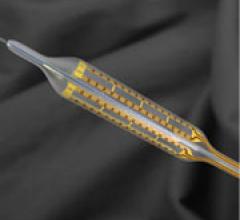
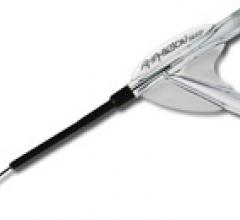
The focus on balloon angioplasty largely slipped from the interventional limelight a decade ago after the ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
June 11, 2009 - Cook Medical announced today the completion of patient enrollment in its REFORM clinical trial ...
June 09, 2009 – Talecris Biotherapeutics Inc., said today it was granted orphan drug designation by the FDA for ...
June 9, 2009 - AngioScore Inc. said today its flagship product, the AngioSculpt Scoring Balloon Catheter, received ...
June 2, 2009 – During their report Friday at the 63rd Annual Meeting of the Society for Vascular Surgery ...
May 8, 2009 - Preliminary data presented yesterday at the 32nd annual SCAI Scientific Sessions show that 82 ...
May 6, 2009 - Spectranetics Corp. yesterday said it made a pre-IDE (investigational device exemption) submission ...
April 16, 2009 - Minnow Medical this week received the CE mark, clearing its disposable Guided Reshaping ...
April 10, 2009 — Invatec has entered into a strategic distribution agreement with Cardiovascular Systems Inc. (CSI ...
April 2, 2009 — Abbott yesterday announced the initiation of the MOBILITY clinical trial to study the safety and ...
April 2, 2009 - TCA Cellular Therapy’s Medical Director Gabriel Lasala, M.D. presented preliminary results of an ...
April 2, 2009 – Invatec received 510(k) clearance from the FDA to market its Amphirion Deep 150 mm Long PTA ...
March 19, 2009 - The FDA has approved an investigational device exemption (IDE) for a multicenter clinical trial ...
March 18, 2009 - Flexible Stenting Solutions Inc. (FSS) this week submitted an investigational device exemption ...
March 10, 2009 – As early detection increases for peripheral arterial disease (PAD) and more patients seek ...

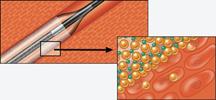
 June 23, 2009
June 23, 2009