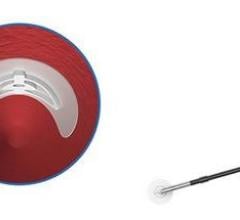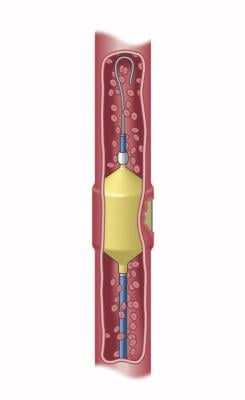
March 29, 2022 – Artio Medical, Inc., a medical device company developing innovative products for the peripheral vascular, neurovascular, and cardiology markets, has received US Food and Drug Administration (FDA) clearance for its Solus Gold Embolization Device, a next-generation product for peripheral vascular occlusion.
"Current occlusion devices can be difficult to position in challenging anatomy, often require multiple implants, and usually take time to completely occlude the target vessel. This can be a problem when physicians need to control or prevent bleeding and reduce the risk of further complications," commented Dr. Blaise Baxter, Vice President of Neurovascular and Peripheral Vascular Interventions for Artio Medical. "The Solus Gold device aims to overcome these limitations with a single implant designed for precision placement and immediate occlusion, even in high-flow vessels and short landing zones."
The Solus Gold Embolization Device is indicated to obstruct or reduce the rate of blood flow in the peripheral vasculature. The delivery system balances flexibility and pushability, enabling physicians to navigate tortuous vasculature. Following a controlled expansion and simple mechanical detachment, the non-porous, balloon-like gold implant provides 360° vessel apposition for immediate and complete occlusion, and resists migration and recanalization, providing physicians with a "one-and-done" solution for peripheral vascular occlusion.1,2 As demonstrated in a GLP pre-clinical study, the Solus Gold device achieved immediate occlusion in > 90% of cases, including in large and high flow vessels, and demonstrated 0% migration and 0% recanalization in testing out to 3 months.1
"We would like to thank the FDA for providing helpful input to the Artio team through the Q-Submission process, and for the careful and thoughtful review of our 510(k) application. I would also like to thank the talented and dedicated Solus Gold product development team at Artio, our many partners in the industry, and our supportive investors for helping Artio reach this important milestone," said F. Nicholas Franano, MD, President and CEO of Artio Medical. "When I first learned to embolize vessels using small metal coils more than 20 years ago, I thought surely someone will come up with a better device for doing that. As we prepare for the market release of the Solus Gold device later this year, I am excited to have the opportunity to provide physicians and patients with an improved peripheral vascular embolization experience."
For more information: https://artiomedical.com/
Reference:
1. Artio Medical Pre-Clinical Study MAE006, performed per TP0397 Rev. B – pre-clinical GLP study. Solus Gold (n=10); MVP (n=10).
2. Artio Medical Pre-Clinical Studies: MAE003-ISO4 (Solus Flex Embolization Device; Medtronic MVP; Cook Nester Coils), GNA1701W (Abbott Vascular AVP II; Abbott Vascular AVP 4), GNA1301W (Abbott Vascular AVP II), GNA1501W (Cook Nester Coils). 2015–2019.

 October 07, 2025
October 07, 2025