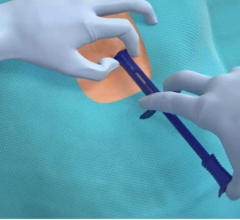
January 20, 2017 — Biotronik has enrolled the first patients in the BioInsight clinical study evaluating the safety and feasibility of performing the minimally invasive BioMonitor 2 insertion procedure in an office setting.
BioMonitor 2 is an insertable cardiac remote monitor with ProMRI technology that is placed underneath a patient's skin to help physicians accurately detect and diagnose atrial fibrillation and syncope (fainting). Atrial fibrillation is a leading cause of stroke and heart failure. BioMonitor 2 provides the highest signal amplitude on the market, according to Biotronik, which leads to excellent sensitivity for improved reporting accuracy. The device can also be used to monitor atrial fibrillation in patients who have undergone ablation procedures. More than 2,000 BioMonitor 2 devices have been sold in the United States since U.S. Food and Drug Administration (FDA) approval in April 2016.
"One of the benefits of BioMonitor 2 is the ease of the insertion procedure, which typically only takes a few minutes," commented Raul Weiss, M.D., cardiologist at Ohio State University. "In-office procedures can reduce patient's and physician's time, increase access to the device and reduce cost burdens for healthcare systems. Early insertion will likely also reduce the time needed for a conclusive diagnosis."
The BioInsight study is a multi-center, prospective, non-randomized post-market study. Participants will receive Biotronik's BioMonitor 2 via in-office insertion and will be evaluated for 90 days to monitor for any potential adverse events, including infection and bleeding.
The BioInsight study is expected to be completed in the third quarter of 2017.
For more information: www.biotronik.com


 April 14, 2026
April 14, 2026