As technology continues to advance for all diagnostic imaging modalities, it sometimes reminds me of a race between vendors to build a better mouse trap. The main issue between cardiac echo, computed tomography (CT), magnetic resonance imaging (MRI), nuclear (SPECT and PET) and invasive angiography is that each has its strengths and weaknesses. When one test falls short, another is used to add diagnostic information, but it also adds time and cost. Unfortunately, in this era of cutting costs and bundled payments, the holy grail of medical imaging would be a one-stop shop cardiac imaging test that combines the majority of the key assets of each of these modalities.
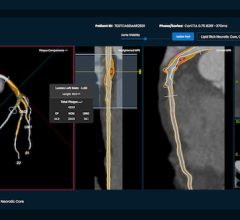
I attended the Society of Cardiovascular Computed Tomography (SCCT) annual meeting in July, where both cardiologists and radiologists came together to make a case for why CT will become the primary cardiac imaging test in coming years. It showed CT radiation is declining and new technology can bring cardiac CT doses below 1 mSv, which is key to CT’s wider acceptance. Data from several trials demonstrate the viability of CT to accurately show myocardial perfusion and fractional flow reserve (FFR) data for the entire coronary tree from a single scan. Work is also continuing to improve plaque characterization software to bring it on par with invasive catheter-based virtual histology intravascular ultrasound (VH-IVUS) or spectrographic plaque analysis. Additionally, spectral CT imaging may enable a new way to image chemical makeup of tissue and allow new types of contrast media.
Speakers at SCCT discussed the use of gold nanoparticles with a polyethylene glycol (PEG) coating, or tantalum, as replacement agents for iodine blood imaging; bromide and fluorine with a phospholipid coating to image liver tumors; and iodine-loaded or bismuth nanoparticles with a biomarker to target fibrin to clearly image blood clots. Nanoparticles may also boost the ability of iodine as a contrast agent. By coating iodine inside liposome particles, the outer shell will protect the iodine from breaking down immediately, both extending its imaging half-life and possibly aiding renal clearance over a longer period of time to reduce contrast-induced nephropathy (CIN).
New types of contrast, especially if combined with spectral CT, may enable inflammation and macrophage imaging, both components of vulnerable plaque. If validated, this new way to image vulnerable plaques prior to rupture might offer a novel way to determine heart attack risk and possibly identify lesions to stent before a heart attack occurs.
Watch a video interview regarding these advances in cardiac CT.



 April 15, 2026
April 15, 2026