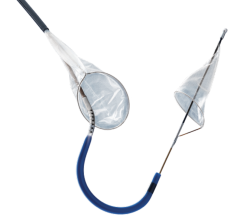
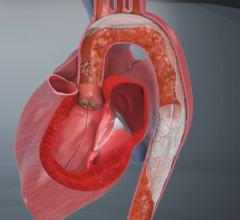
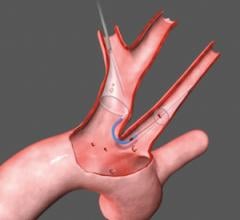
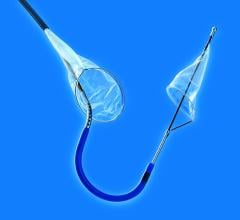
November 1, 2011 – Claret Medical Inc. said its Montage System has received CE mark approval as the world's first complete system of filtration to protect both carotid arteries simultaneously. The 6 French catheter is delivered via radial or brachial access, providing complete neuro protection during intravascular procedures such as transcatheter aortic valve intervention (TAVI). The unique design of the Montage System not only provides neuro-protection from debris, but also captures and removes potential stroke-causing embolic material from the body.
"The CE mark approval for the Montage System is an exciting new step forward to potentially reduce stroke rates in TAVI and other vascular procedures," said Prof. Eberhard Grube, M.D., FACC, FSCAI, director of the structural heart program at University Hospital in Bonn, Germany. "The timing of this innovation could not be better, in that it has the potential to change the way physicians view TAVI in their practice."
"The Montage System was surprisingly elegant and simple to deliver," said Alexander Abizaid, director, Catheterization Laboratory, Institute Dante Pazzanese, Sao Paolo, Brazil. "We, along with other investigators, were astounded at the volume of debris we found in the filters and were delighted we could provide a solution to help capture and remove this material from the patient's vascular circulation."
This is the second product approval for Claret Medical in 2011. These approvals were supported by data from the clinical trial in Europe, which included 30-day patient follow-up. To further study the device in an expanded population, Claret plans to initiate additional controlled clinical trials outside of the United States early next year.
For more information: www.claretmedical.com


 April 25, 2023
April 25, 2023