August 5, 2016 — The Centers for Medicare and Medicaid Services (CMS) has reassigned MitraClip transcatheter mitral valve repair procedures to the diagnosis-related group (DRG) codes 228-229 for “Other Cardiothoracic Procedures, with and without major complications or comorbidities (MCCs)." The change is expected to boost reimbursement and allow more Medicare patients to have access to the technology.
“We commend CMS for this final step to ensure adequate reimbursement for MitraClip,” Abbott said in a statement. “With this rule, CMS has opened the door for Medicare patients who may benefit from MitraClip to have broader access to the device. We appreciate CMS’s proactive approach in recognition of the substantial benefits of this therapy — from issuing a National Coverage Determination, New Technology Add-on Payment approval, physician reimbursement and now MS-DRG reassignment that will make hospital MitraClip programs sustainable.”
Abbott said the changes demonstrate the government’s commitment to provide access to innovation for seniors and others in the Medicare program.
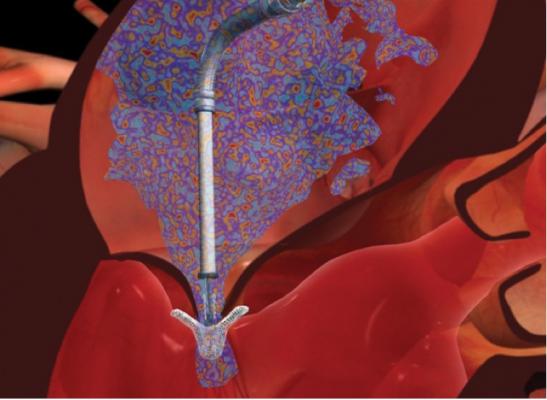
MitraClip is the only FDA-cleared treatment of its kind for percutaneous mitral valve repair. It is a performed as a beating-heart procedure with no cardiopulmonary bypass required. It allows for real-time positioning and repositioning to optimize mitral regurgitation reduction, and is designed to preserve surgical options.
The ruling can be viewed at www.federalregister.gov/public-inspection.
Watch the video "Transcatheter Mitral Valve Repair Technologies."


 April 10, 2026
April 10, 2026 









