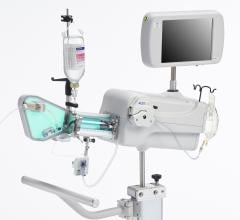
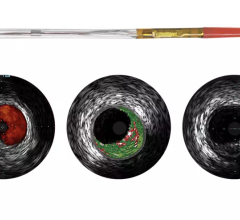
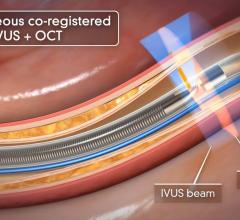
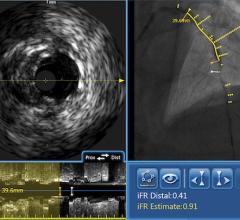
May 27, 2010 – A single catheter combining both intravascular ultrasound (IVUS) and near-infrared (NIR) spectroscopy to identify lipid-core plaques (LCP) was unveiled at EuroPRC in Paris this week. The device also was demonstrated in live patient cases.
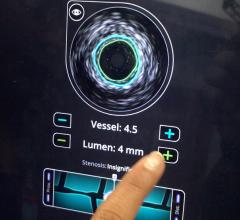
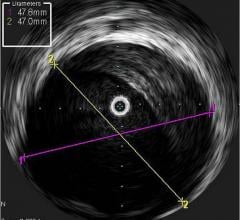
The LipiScan IVUS Coronary Imaging System is designed to identify and characterize diseased coronary tissue and guide therapeutic interventions
As a highlight of EuroPCR's "Glimpse into the Future" session held today, a live case demonstration of the LipiScan IVUS system was broadcast from the Thoraxcenter of the Erasmus University Medical Center in Rotterdam, under the direction of professor Willem J. van der Giessen and Dr. Nicolas Van Mieghem.
InfraReDx developed the system in collaboration with clinicians and researchers at the biomedical engineering department of the Thoraxcenter. It is a one-catheter, single-pullback system and the only coronary imaging device known to combine both IVUS technology and NIR spectroscopy.
Lipid-core containing plaque is believed to be "vulnerable plaque" that can rupture and form dangerous blood clots, leading to coronary thrombosis. LCP is widely considered to be responsible for a significant number of stenting procedure complications, and is believed to be the primary cause of acute coronary events, the leading cause of death in the developed world.
The LipiScan IVUS system is expected to become commercially available in the U.S. during the second half of 2010 and in Europe during 2011. InfraReDx currently markets its first-generation LipiScan NIR system, which is the only FDA-cleared product to incorporate near-infrared imaging to identify and assess the chemical composition of coronary plaque, particularly LCP.
The addition of complementary IVUS imaging technology to the LipiScan platform enhances its capabilities by also allowing for structural assessment of coronary plaque. LipiScan IVUS was designed to provide physicians with a one-stop determination of critical coronary health factors that guide patient care, including: chemical composition, degree of stenosis, stent expansion, plaque burden, estimation of cap thickness and extent of positive remodeling.
"The interventional cardiology community attending EuroPCR will be looking with great interest at the LipiScan IVUS system, as it represents an important technological advance in patient care," said Evelyn Regar, M.D., Ph.D., interventional cardiologist at the Thoraxcenter and co-chairperson for the EuroPCR session. "The ability to at once assess the presence, makeup and structure of lipid-core vulnerable plaques would be of great utility during stenting procedures. It is our hope and expectation that the improved imaging and diagnostic capabilities offered by the LipiScan IVUS system will result in reduced stenting complications and better informed treatment decisions for patients with coronary artery disease."
"I am eager to view the live case demonstration of LipiScan IVUS," said James Goldstein, M.D., director of cardiac research and education for Beaumont Hospital in Michigan. "The first-generation LipiScan NIR system is already in use at our hospital, and its chemogram information has already begun providing very promising results. The addition of structural IVUS data via a single catheter and in a single pull-back procedure would further elevate the system's practical clinical utility for characterizing coronary lesions to reduce stenting complications and better guide cardiac care."
Goldstein, who has been involved in vulnerable plaque related research for over 10 years, currently serves as a consultant to InfraReDx as well as other medical device and imaging technology companies.
For more information: www.infraredx.com


 September 18, 2025
September 18, 2025