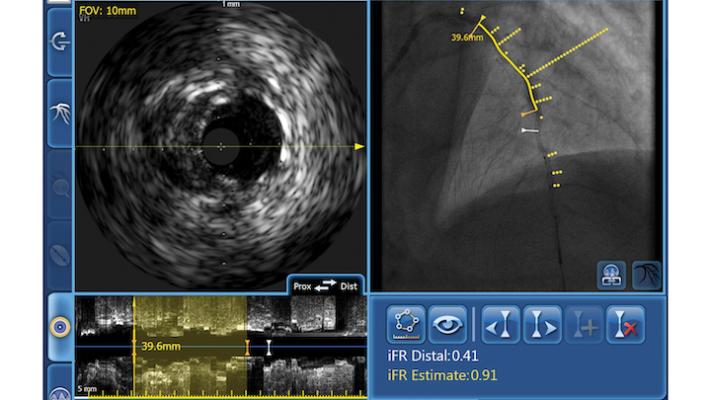
Example of Philips' IVUS system, which can be co-registered with angiography and FFR to offer more information on coronary lesions.
Intravascular ultrasound (IVUS) is the workhorse intravascular imaging modality in cardiac cath labs. It is used to help decide the best therapy option, guide pre-stent planning and then confirm and optimize treatment results. This confirmation is mainly determining if a deployed stent is fully apposed to the vessel wall, as exposed stent struts can be a cause of restenosis.
While the competitive technology of optical coherence tomography (OCT) offers much higher definition imaging, the reflected light-based technology lacks the tissue penetration of ultrasound, which can see deeper into vessel walls and thicker plaques. OCT is also more technically challenging, because it requires the blood pool in a vessel segment to be cleared using saline or contrast so the light emitted from the catheter can penetrate tissue and reflect back without interference from the opaque blood, which would mask the signal.
IVUS uses a miniaturized ultrasound transducer mounted on the tip of a catheter. It captures real-time, high-resolution images inside the vessel. It can image the current lumen, plaques and vessel walls showing the true lumen under the plaque. This can be used for measurements for the length of a coronary lesion and the actual diameter of the vessel for proper stent sizing and to identify the best landing zone for the stent. IVUS can aid if interpreting vessel obstructions seen on angiography by helping interpret vessel morphologies. It is often used to help get a clearer understanding because of ambiguous angio imaging, postal lesions, bifurcation lesions, diffuse disease, and in repeat patients and lesions in the left main.
Boston Scientific was first to market with this technology, followed by Volcano (now part of Philips Healthcare), Acist, InfraredX and Conavi.
Newer IVUS Systems
Philips has advanced IVUS in recent years, enabling co-registration with both angiography and fractional flow reserve or instant wave-free ratio (FFR/iFR) physiological assessments. This co-registered view can be displayed on the overhead screen in the lab, helping add more information about lesions and their severity in one location. It also helps quickly identify which is the culprit lesion in heavily disease arteries.
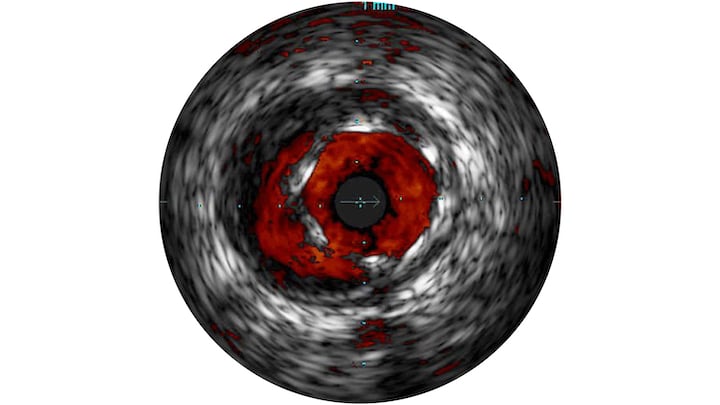
The Philips system also offers ChromaFlow, which is similar to Doppler ultrasound, showing blood flow within the vessel in red. This can help make the grainy black and white IVUS images easier to interpret by clearly showing the vessel lumen, or the junction of a bifurcating vessel.
Philips also offers an array of seven different IVUS catheters in its portfolio aimed at both the coronary and peripheral vessels with different degrees of functionality.
In 2018, Conavi Medical received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Novasight Hybrid OCT/IVUS System. The hybrid system provides the excellent image resolution and contrast of OCT, but still offers the larger field of view with simultaneous IVUS. The system displays the combined images in a precise, co-registered fashion.
Near Infrared Spectroscopy IVUS Aids in Detecting Vulnerable Plaques
Infraredx, a Nipro Company, developed near infrared spectroscopy (NIRS) a decade ago as a completing intravascular imaging modality to image areas of high lipid burden in vessel segments. However, the imaging was difficult for most interventionists to interpret. When IVUS was added to the system a few years later; the imaging system offered a new way to look inside coronary vessels and to analyze the plaque composition.
Many interventional experts immediately saw the opportunity to use the system as a vulnerable plaque detector to pinpoint lipid rich plaques (LRP) to monitor closely or to possibly pre-emptively stent before a cardiac event occurs. The LRP and PROSPECT II studies both demonstrated NIRS-IVUS can help identify angiographically non-obstructive lesions with high-risk characteristics for future adverse cardiac outcomes.[1,2]
“Multi-vessel NIRS can be easily and safely performed to assess and identify vulnerable patients and vulnerable plaques,” said Ron Waksman, M.D., associate director of the Division of Cardiology at MedStar Heart Institute in Washington, D.C. “Intravascular NIRS imaging in mildly or non-obstructive coronary arteries can be used as a tool to identify both patients and nonculprit arteries at high risk for future events and should be considered for use in patients undergoing cardiac catheterization with possible PCI.”
Watch the VIDEO: Near Infrared Spectroscopy Detects, Aids Risk Assessment of Vulnerable Plaques — interview with Waksman on this technology.
The latest version of the Infrared system, the Makoto, automatically quantifies the total lipid core in regions of interest as the lipid core burden Index (LCBI). By analyzing both structural and compositional data with NIRS-IVUS, the effects of medical treatments on plaque size and lipid core content can be assessed. The technology is being used in research to develop novel preventive approaches for plaque stabilization and to study the regression of plaque size and composition.
IVUS For Peripheral Vessels and Veins
Intravascular imaging has been used primarily for coronary arteries, but is finding utility in for interventional procedures in both peripheral arteries and veins.
In the realm of peripheral arteries, there has been no consensus on the use of IVUS until October 2020, when a worldwide committee of 40 cross-specialty medical experts created the first consensus for the appropriate use of IVUS in peripheral vascular disease (PVD) interventions. Today, healthcare providers’ use of IVUS in PVD interventions is not standardized and is therefore inconsistent. The new appropriate-use expert consensus aims to help establish global standards of care to adopt into guidelines and improve quality care in PVD.
The new consensus and recommendations for use of IVUS aim to improve quality care in PVD and are based on evidence, expert practice standards, and clinical experience.
“The results of the cross-specialty expert consensus demonstrate strong support for the use of IVUS during peripheral interventions,” said Eric A. Secemsky, M.D., interventional cardiologist at Beth Israel Deaconess Medical Center and assistant professor of medicine at Harvard Medical School. “Future efforts need to focus on improving IVUS implementation into clinical practice and streamlining procedural workflow to help improve our patient outcomes.”
The consensus document included review of key clinical IVUS scenarios and decision-making processes, and the results were shared at a special symposium during Vascular Interventional Advances (VIVA) 2021. Through this method, experts established clinical consensus to identify optimal use of IVUS and potential gaps to set a standard across clinical specialties and drive positive outcomes for patients.
In venous interventions, one recent study on IVUS was found to be superior to venography in determining the proper location of treatment zones in a blinded comparison study that included large series of iliac vein stent cases. The study included 155 limbs treated for chronic iliac vein occlusion.[3] It found With regards to evaluation of the main venous stenosis, venography failed to identify the stenosis in 19% of cases, underestimated the degree of stenosis and failed to locate accurately the stenosis in 68% of cases. The study found IVUS was more accurate at all three of these key assessments.
“Adequate assessment of the location and degree of stenosis and delineation of venous anatomy for optimal landing zones are key elements in the success of interventions to treat chronic obstructions of the deep venous system,” explained lead author Myriam Montminy, M.D., MSc, FRCSC, from The RANE Center, St. Dominic’s Memorial Hospital, Jackson, Miss. “While venography is more accessible and less expensive to perform than IVUS, an increasing number of studies demonstrate that IVUS is significantly more sensitive than venography in identifying stenotic lesions in the iliac-caval segments. Our study aimed to take this one step further by comparing these modalities in identifying the key parameters required to guide stent placement.”
Related IVUS Technology Content:
Comparison Chart of IVUS Catheters
Comparison Charts for IVUS Systems
10 Reasons Why You Need Supplemental Imaging in the Cath Lab
IVUS Demonstrates Greater Visualization of Dissections in iDissection Classification Study
VIDEO: Near Infrared Spectroscopy Detects, Aids Risk Assessment of Vulnerable Plaques — Interview with Ron Waksman. M.D.
NIRS-IVUS Imaging Can Help Identify High-Risk Plaques That Can Lead to Adverse Outcomes
IVUS-Guided Drug-Eluting Stent Implantation Improves Clinical Outcomes in All-Comer Patients
References:
3. Myriam L. Montminy, James D. Thomasson, Guillermo J. Tanaka, et al. A comparison between intravascular ultrasound and venography in identifying key parameters essential for iliac vein stenting. Journal of Vascular Surgery: Venous and Lymphatic Disorders. November 2019, Volume 7, Issue 6, Pages 801–807. DOI: https://doi.org/10.1016/j.jvsv.2019.03.015.


 May 13, 2026
May 13, 2026 









