October 20, 2021 — Philips, a global leader in health technology and intravascular ultrasound (IVUS) solutions, announced that a worldwide committee of 40 cross-specialty medical experts achieved the first-ever consensus for the appropriate use of IVUS in peripheral vascular disease (PVD) interventions. The new consensus and recommendations for use of IVUS aim to improve quality care in PVD and are based on evidence, expert practice standards, and clinical experience.
“The results of the cross-specialty expert consensus demonstrate strong support for the use of IVUS during peripheral interventions,” said Eric A. Secemsky, M.D., Interventional Cardiologist at Beth Israel Deaconess Medical Center and Assistant Professor of Medicine at Harvard Medical School. “Future efforts need to focus on improving IVUS implementation into clinical practice and streamlining procedural workflow to help improve our patient outcomes.”
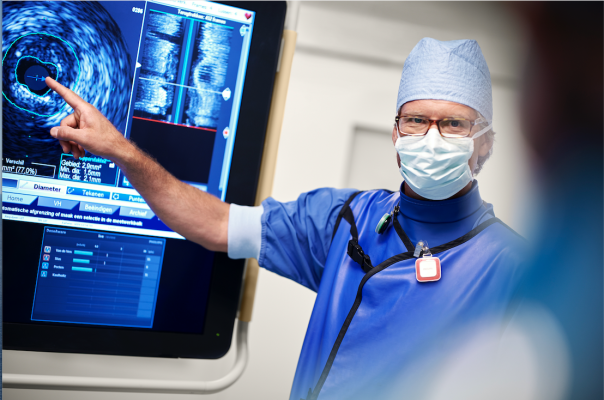
As part of Philips’ peripheral vascular portfolio, IVUS provides the visualization and guidance essential for the optimal diagnosis and treatment of patients. Using a miniaturized ultrasound transducer mounted on the tip of a catheter, it captures real-time, high-resolution images inside the vessel. IVUS imaging enables physicians to formulate patient-specific treatment plans and confirm therapy results.
Today, healthcare providers’ use of IVUS in PVD interventions is not standardized and is therefore inconsistent. The new appropriate-use expert consensus may help establish global standards of care to adopt into guidelines and improve quality care in PVD.
To achieve consensus, the broad, multi-disciplinary, global experts used a rigorous methodology. They conducted a systematic and comprehensive review of key clinical IVUS scenarios and decision-making processes before voting as world experts in the field. The results were shared at a special symposium during Vascular Interventional Advances (VIVA) 2021 (Las Vegas, 5-7 October). Through this method, experts established clinical consensus to identify optimal use of IVUS and potential gaps to set a standard across clinical specialties and drive positive outcomes for patients.
"We commend these global, cross-specialty experts, on their commitment to identifying the appropriate uses of IVUS to standardize procedures in order to improve outcomes for peripheral vascular patients worldwide,” said Chris Landon, senior vice president and general manager image guided therapy devices at Philips. “This expert consensus sets a precedent on how patients are managed and supports Philips’ commitment to evidence-based medical guidelines as part of our pursuit of better patient outcomes, enhanced patient and staff experiences, and lower cost of care.”
For more information: www.philips.com
Related IVUS Technology Content:
Comparison Chart of IVUS Catheters
Comparison Charts for IVUS Systems
10 Reasons Why You Need Supplemental Imaging in the Cath Lab
IVUS Demonstrates Greater Visualization of Dissections in iDissection Classification Study
VIDEO: Near Infrared Spectroscopy Detects, Aids Risk Assessment of Vulnerable Plaques — Interview with Ron Waksman. M.D.
NIRS-IVUS Imaging Can Help Identify High-Risk Plaques That Can Lead to Adverse Outcomes
IVUS-Guided Drug-Eluting Stent Implantation Improves Clinical Outcomes in All-Comer Patients


 November 09, 2025
November 09, 2025