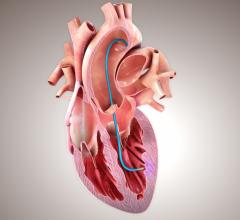
November 18, 2011 – CorMatrix Cardiovascular Inc. announced that the U.S. Food and Drug Administration (FDA) has granted the company full investigational device exemption (IDE) approval for its ongoing multi-center, prospective, randomized clinical trial aiming to demonstrate the safety and efficacy of the CorMatrix ECM for Pericardial Closure to reduce the incidence of new onset postoperative atrial fibrillation following isolated primary coronary artery bypass graft (CABG) surgery. CorMatrix Cardiovascular is a medical device company dedicated to developing and delivering unique extracellular matrix (ECM) biomaterial devices that harness the body's innate ability to repair damaged cardiovascular tissue.
At this time, the clinical trial has enrolled more than 80 percent of the planned 440 total subjects. This prospective trial was initiated based on a previous retrospective study that demonstrated a statistically significant reduction in the rate of new onset postoperative atrial fibrillation in CABG patients following pericardial reconstruction with the CorMatrix ECM. As the current CorMatrix trial moves toward completion, positive results would be yet another milestone determining the role of pericardial reconstruction using the CorMatrix material in cardiovascular surgery.
"We are pleased with the FDA's decision to provide us with full approval for this significant trial, and we are on track to complete enrollment in the study, analyze the results and submit them for publication by the end of next year," said Robert G. Matheny, M.D., chief scientific officer, CorMatrix Cardiovascular. "We continue to encounter data from the clinical community suggesting an increase in observed postoperative atrial fibrillation and the significant impact both clinically and economically to the healthcare system. This trial will be important to evaluate if our CorMatrix implant can foster better outcomes in this subset of at-risk patients."
Following cardiac surgery, new onset postoperative atrial fibrillation is the most common arrhythmic complication, with reported incidence between 32 percent and 64 percent, posing a serious concern for patients undergoing cardiac surgery procedures [Creswell 1993; Auer 2005; Echahidi 2008]. A recent retrospective analysis of more than 16,000 patients with no history of atrial fibrillation who underwent CABG surgery found an association between new onset postoperative atrial fibrillation and a 21 percent relative increase in mortality, with the greatest negative impact seen in the long-term survival of women [El-Chami 2010].
"Trials designed to test preventative measures for postoperative atrial fibrillation are not only interesting, but they are necessary given the need to improve outcomes and create more effective and cost-efficient models of healthcare," added James L. Cox, M.D., Emeritus Evarts A. Graham Professor of Surgery, Washington University School of Medicine, Barnes-Jewish Hospital, St. Louis, Mo., president and CEO, World Heart Foundation, and CorMatrix Scientific Advisory Board member. "Medical advancements to reduce postoperative arrhythmias have not materialized, and we continue to see an increase in the number of patients encountering these symptoms. With bypass surgery continuing to be an unfortunately common procedure, it is imperative that surgeons are given options to help improve patient care and reduce unwanted outcomes, while reducing the overall cost-burden to the healthcare system."
About Extracellular Matrix Biomaterial
The unique properties of extracellular matrix biomaterials were discovered at Purdue University. The decellularized matrix material serves as a scaffold to allow adjacent tissues to deliver cells and nutrients to the matrix, which then differentiate into tissue-specific cells. The ECM material is gradually replaced as the patient's own body reinforces and rebuilds the weakened site. During the repair, the matrix is naturally degraded and resorbed, leaving remodeled functional tissue where scar tissue or injured tissue would normally be expected.
The use of extracellular matrix materials in non-cardiovascular applications has established a significant foothold in soft tissue repair, wound management and orthopedic applications. The safety of extracellular matrices has been well established in a number of different clinical applications. The extracellular matrix has been studied extensively, with more than 500 published papers. Since 1999, an estimated one million patients worldwide have received an extracellular matrix implant.
For more information: www.cormatrix.com


 November 19, 2021
November 19, 2021