
An example of cardiac ultrasound longitudinal strain measurements on the Epsilon EchoInsight software. This can be used to assess cardiotoxicity from chemotherapy agents.
February 25, 2020 — Results of a multi-centre, international, clinical trial co-led by Peter Munk Cardiac Centre (PMCC) cardiologist Dinesh Thavendiranathan, M.D., point to the benefit of using a more sensitive cardiac ultrasound test to detect cardiotoxicity heart function issues early, so cancer patients do not have to fight heart failure too.
Unfortunately, for 1 in 20 high-risk patients, treating cancer with certain therapies means the added potential of developing heart failure. The one-year results of the highly anticipated trial compared heart function at the end of anthracycline-based chemotherapy, a treatment that can successfully treat cancer but also damage the heart, known as cardiotoxicity.
“When I was diagnosed, I was scared for my life,” said Sindhu Johnson, a breast cancer patient and one of 307 participants, 90% of which had breast cancer, enrolled in the trial. “I was willing to undertake any treatment, but I understood my survival may come at a price. And that price included cardiotoxicity.”
Thavendiranathan is the lead of the Cardiotoxicity Prevention Program at the Ted Rogers Centre for Heart Research at the PMCC and a recognized leader in cardiotoxicity.
“We hoped our study would show a better way to care for cancer patients, who are already fighting one disease and should not have to worry about the future risk of heart failure too,” Thavendiranathan explained.
Trial participants were split into two groups and monitored during their cancer treatment. One group received the current standard of care, measuring Left Ventricular Ejection Fraction (LVEF) with traditional imaging technology known as echocardiography. The other group received the current standard of care as well as an additional imaging method, measuring Global Longitudinal Strain (GLS).
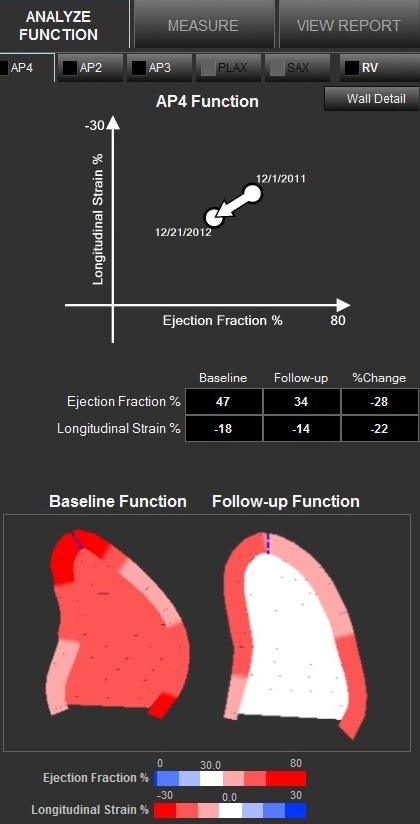 LVEF measures how much blood is pumped out of the left ventricle of the heart with each contraction, whereas GLS measures deformation of the heart muscle.
LVEF measures how much blood is pumped out of the left ventricle of the heart with each contraction, whereas GLS measures deformation of the heart muscle.
“LVEF is the traditional imaging method, but we already know measuring GLS allows for a better identification of heart damage,” said Thavendiranathan. “What we didn’t know is if we can reduce the risk of cardiotoxicity by intervening once this early heart damage is detected.”
Published in the Journal of the American College of Cardiology, the Strain Surveillance of Chemotherapy for Improving Cardiovascular Outcomes (SUCCOUR) randomized controlled trial did not meet its primary outcome.[1] There was no difference in the continuous measure of heart function for all patients in the LVEF-guided and GLS-guided arms in both treated and untreated patients.
However, what is of clinical importance are the patients whose abnormal change in heart function resulted in the initiation of therapy.
In these patients, the use of GLS-monitoring led to a lower reduction in heart function versus traditional monitoring. It also reduced the risk of cardiotoxicity, and heart damage was detected earlier and in twice the number of participants.
For oncologists and cardiologists, one of the primary concerns is how to prevent the development of cardiotoxicity in patients. The secondary outcome of the SUCCOUR trial proves by using GLS to monitor heart function and intervening with cardioprotective therapy when damage is detected, further reduction in heart function can be prevented and the risk of cardiotoxicity reduced.
“The purpose of a sensitive method like GLS is to pick up the presence of disease and treat it early,” said Thavendiranathan. “This means more patients will be treated, and if we start heart medications when a change is identified, we can prevent significant worsening of heart function.”
Sindhu, who was randomized into the GLS arm, joined the SUCCOUR trial knowing it may reduce the degree of cardiotoxicity she could potentially develop.
When a change in measurement alerted Thavendiranathan that the cancer therapy was damaging her heart, he initiated cardio-protective therapy — drugs that help improve heart function and prevent further damage.
By the end of her treatment, Sindhu’s heart function returned to normal and she did not develop cardiotoxicity.
“Our findings suggest we should consider changing how we follow patients during cancer therapy and add GLS to routine heart surveillance,” said Thavendiranathan, who co-led the study at 28 centers with Dr. Thomas Marwick of the Baker Heart and Diabetes Institute, and Dr. Tomoko Negishi of the Menzies Research Institute.
Thavendiranathan would like to see the GLS-measurement approach implemented in clinical practice.
“This trial gave me hope, and I’m thankful to say I’m now doing well and the cancer treatment worked,” said Sindhu. “Dr. Thavendiranathan is an excellent physician and researcher, and I’m very thankful to him.”
Thavendiranathan is supported by the Ted Rogers Centre for Heart Research and other donors to Toronto General and Western Hospital Foundation, the Canadian Institutes of Health Research New Investigator Award, the Ontario Early Research Award, and a Canada research chair in cardio-oncology.
The SUCCOUR trial is made possible through the support of the Princess Margaret Cancer Centre at the University Health Network.
For more information: www.petermunkcardiaccentre.ca
Related Chemotherapy Cardiotoxicity Content:
Strain Imaging Improves Cardiac Surveillance of Certain Breast Cancer Patients
VIDEO: Assessing Cardiotoxicity Response With Cardio-Oncology Echo Imaging — Interview with Federico Asch, M.D.
Assessing Cardiotoxicity Due to Cancer Therapy
VIDEO: An Overview of Echo Strain Imaging — Interview with Marielle Scherrer-Crosbie, M.D.
VIDEO: Linking Breast Cancer Treatment to Cardiac Issues
AHA Puts New Focus on Where Heart Disease and Breast Cancer Treatment Meet
VIDEO: Characterization of Cardiac Structural Changes and Function Following Radiation Therapy — Interview with Magid Awadalla, MBBS


 May 06, 2026
May 06, 2026 









