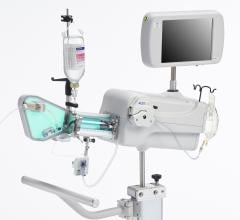
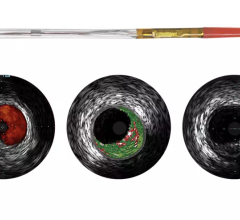
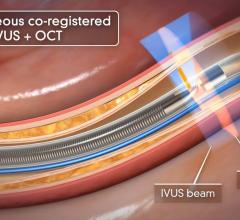
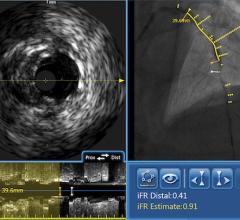
June 28, 2012 — Infraredx Inc., a medical device company committed to advancing the diagnosis and management of coronary artery and other vascular diseases, today announced that the company has signed an exclusive five-year agreement with Nipro Corporation to distribute the TVC Imaging System for true vessel characterization of coronary artery disease in Japan. This agreement expands Infraredx’s presence into Asia and demonstrates the company’s commitment to make the TVC Imaging System available worldwide. The TVC Imaging System is an intravascular imaging system that integrates near infrared spectroscopy (NIRS) lipid core plaque (LCP) detection and enhanced intravascular ultrasound (IVUS) imaging technology to assess vessel composition and structure.
Under the terms of the agreement, Infraredx will receive a minimum of $50 million in revenues over the duration of the agreement. Nipro will be responsible for obtaining marketing authorization for the TVC Imaging System and catheter with Japan’s Pharmaceuticals and Medical Device Agency (PMDA). Additionally, Nipro will be responsible for all sales, marketing and promotion of the TVC System, including clinical education and physician training in the operation of the TVC system as well as all field service and technical support of the TVC System. The agreement is contingent upon a Shonin (product approval) by the Japanese regulatory authorities. The companies anticipate approval and subsequent launch of the TVC Imaging System in Japan in 2013.
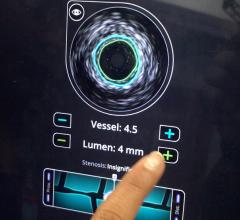
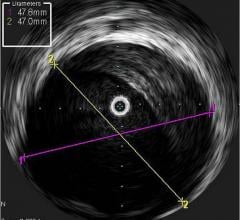
IVUS has become the standard of care in Japan with more than 80 percent of all percutaneous coronary intervention (PCI) procedures guided by intravascular imaging. However, while IVUS can display the structure of a plaque, it is not an optimal method to determine its composition and if it contains a lipid core. Lipid core plaque is a fatty coronary artery plaque suspected to be vulnerable to rupture and cause most heart attacks. It is also known to complicate stenting procedures either during or following the placement of coronary artery stents. The TVC Imaging System is the only device that offers physicians both IVUS and NIRS to improve characterization of the vessel.
For more information: www.infraredx.com


 September 18, 2025
September 18, 2025