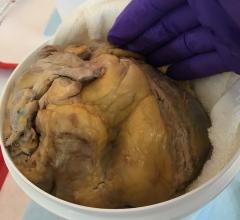
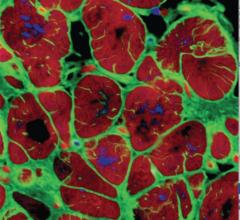
NRG1 injections improve cardiac function after MI through cardiomyocyte proliferation. Representative examples are shown from each group scar blue and myocardium is red.
July 24, 2009 – Injured heart tissue normally can't regrow, but researchers at Children's Hospital Boston have now laid the groundwork for regenerating heart tissue after a heart attack without the use of stem cells, in patients with heart failure, or in children with congenital heart defects.
In the July 24 issue of Cell, they show that a growth factor called neuregulin1 (NRG1), which is involved in the initial development of the heart and nervous system, can spur heart-muscle growth and recovery of cardiac function when injected systemically into animals after a heart attack.
After birth, heart-muscle cells (cardiomyocytes) normally withdraw from the cell cycle – meaning they stop dividing and proliferating. But the researchers, led by Bernhard Kuhn, M.D., and Kevin Bersell of the department of cardiology at Children's, were able to restart the cell cycle with NRG1, stimulating cardiomyocytes to divide and make copies of themselves – even though they are not stem cells.
"Although many efforts have focused on stem-cell based strategies, our work suggests that stem cells aren't required and that stimulating differentiated cardiomyocytes to proliferate may be a viable alternative," says Kuhn, the study's senior investigator and a practicing pediatric cardiologist at Children's.
When the team injected NRG1 into the peritoneal cavity of live mice after a heart attack, once daily for 12 weeks, heart regeneration was increased and pumping function (ejection fraction, assessed on echocardiograms) improved as compared with untreated controls. The NRG1-injected mice also lacked the left-ventricular dilation and cardiac hypertrophy that typify heart failure; both were seen in the controls.
When the researchers also stimulated production of a cellular receptor for NRG1, known as ErbB4, cardiomyocyte proliferation was further enhanced, demonstrating that NRG1 works by stimulating this receptor. They also identified the specific kinds of cardiomyocytes (mononucleated) that are most likely to respond to treatment.
In 2007, Kuhn and colleagues first demonstrated that the heart has dormant regenerative capacities that can be reawakened. Kuhn developed a sponge-like patch, soaked in a compound called periostin that is abundant in the developing fetal heart (and in injured skeletal muscle) but scarce in adult hearts. When the patch was placed over the site of cardiac injury in rats, it induced cardiomyocyte proliferation and improved heart function (Nature Medicine 2007; 13:962-9). Similar results were seen in larger animals, and periostin is now in preclinical development at Children's Hospital Boston for future application in human patients with heart failure.
The new work adds a second compound to the heart-regeneration toolbox, and reveals how both periostin and NRG1 work at the cellular and molecular level, an essential step in predicting possible side effects. Both compounds ultimately act on the same cellular pathway, Kuhn found.
"We applied periostin locally at the site of cardiac injury, but NRG1 works when given by systemic injection – a very promising result that suggests it may be feasible to use this in the clinic to treat heart failure," Kuhn said.
The study was funded by the Department of Cardiology at Children's Hospital Boston, the Charles Hood Foundation, and the American Heart Association.
For more information: www.childrenshospital.org


 November 19, 2021
November 19, 2021