Image courtesy of Covidien
January 8, 2015 — Medtronic's roughly $43 billion acquisition and merger with Covidien has been widely discussed in the media for its U.S. Federal tax implications, but it's also creating a stronger leading player in one of the fastest growing device markets, according to Kalorama Information. The healthcare market research firm said the two companies compete in patient monitoring systems will feature a diverse set of solutions spanning inpatient to home care, and cardiac to respiratory and blood-based monitoring.
Vendor product overviews, market segmentation and market share analysis within advanced remote patient monitoring is available from Kalorama Information in its latest report: Advanced Remote Patient Monitoring Systems, 7th Ed.
The remote patient monitoring market includes wireless and remote devices and technologies that monitor and transmit data through an internal network or secure wireless transmissions. The market includes the devices and associated systems to deliver current or real-time patient data outside of the patient room or from the home.
A core patient monitoring product for Covidien is its Vital Sync virtual patient monitoring system that provides integrated ventilator, pulse oximeter and capnography data to a central station or other access point for clinicians. Other Covidien patient monitoring products are in the respiratory function market segment and include Capnostream (portable and beside capnography or respiratory carbon dioxide monitors), Nellcore (pulse oximetry devices) and BIS monitors (measures patient consciousness under anesthesia).
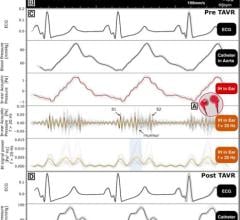
Medtronic's strengths in advanced remote patient monitoring relate to the transmission and management of implantable and on-patient device-generated data. Medtronic's CareLink Network is able to deliver data from implantable pacemakers and defibrillators with cardiac resynchronization features, insulin pumps and continuous glucose monitoring devices. The devices and systems are relevant to the cardiac event and function and blood management and function (diabetes monitoring) market segments under advanced remote patient monitoring.
For more information: www.medtronic.com, www.covidien.com, www.KaloramaInformation.com


 October 21, 2025
October 21, 2025