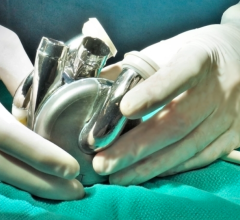
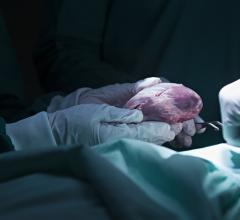
March 24, 2016 — A new documentary produced by Retro Report for The New York Times looks at the history and modern use of the SynCardia temporary Total Artificial Heart (TAH-t). The 12-minute film, “A Change of Heart,” re-examines this leading story of the past and brings viewers through to today’s news.
This documentary details the development of the SynCardia Total Artificial Heart and its direct predecessor, the Jarvik 7. Like a heart transplant, the TAH-t is the only approved device that eliminates end-stage biventricular (both sides) heart failure. This fatal condition results when the native heart's two ventricles can no longer pump enough blood for a patient to survive.
Through personal stories of surgeons and SynCardia Total Artificial Heart patients, this documentary takes the audience from the first use of a Total Artificial Heart for permanent use in 1982 to today's use as a bridge to a donor heart transplant.
"Use of SynCardia's Total Artificial Heart Technology has gone full circle," said Michael P. Garippa, SynCardia's CEO and president. "The first implants were for permanent use. For the last 30 years the TAH-t has been used as a bridge to transplant. Now we are in an FDA [U.S. Food and Drug Administration] Investigational Device Exemption (IDE) clinical study for 19 destination therapy patients who do not qualify for a donor heart transplant."
"This study is generating data on the effective use of the SynCardia Heart as a way for patients to recover from heart failure and continue with a near-normal lifestyle without a human heart," Garippa said.
The 70cc SynCardia Total Artificial Heart, when used for destination therapy, is an investigational device, limited by United States law to investigational use.
For more information: www.syncardia.com


 April 28, 2026
April 28, 2026