November 7, 2007 – Injecting patients’ own stem cells – ones that exist in skeletal muscle – directly into the heart with a three-dimensional guided catheter is feasible, safe and might be superior to optimal medical therapy (OMT), researchers reported in a late-breaking special session on stem cell research at the American Heart Association Scientific Sessions 2007.
Researchers directed the Catheter-Based Delivery of Autologous Skeletal Myoblasts for Ischemic Cardiomyopathy: Feasibility, Safety and Improvement in Cardiac Performance (CAuSMIC) trial to evaluate the effect of adding autologous skeletal myoblasts (ASM) transplantation to Optimal Medical Treatment (OMT), compared to OMT alone.
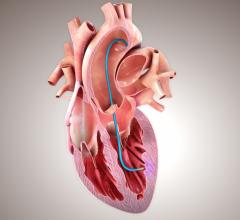
In the randomized, controlled open-label study, the research team gathered cells from patients’ thigh muscles, removing a piece of tissue about the size of a quarter and separating the stem cells. These cells, called myoblasts, were then grown to increase their number and injected into the heart via an investigational catheter using a three-dimensional voltage mapping system “like a small Global Positioning Satellite system,” said Nabil Dib, M.D., director of clinical cardiovascular cell therapy and associate professor of medicine at the University of California in San Diego.
This Phase 1 trial focused on feasibility and safety, but researchers also examined the preliminary efficacy of the procedure. Researchers enrolled 23 heart failure patients whose hearts were pumping inadequately (ejection fraction scores of 40 percent or less). The patients had previously suffered heart attacks and were receiving OMT at the beginning of the trial. Twelve received ASM and OMT and 11 continued receiving OMT alone.
At one year, researchers reported all ASM procedures were successful without injection-related complications, and with no deaths in either group. One person in each group experienced abnormal and rapid heartbeat but both were successfully treated with implantable cardioverter defibrillators. At 12 months follow-up, there were no significant differences in control versus treated patients concerning the occurrence of arrhythmias. ASM-treated patients on average had improved pumping function, reported better quality of life on a questionnaire, and the size of their overworked left ventricles had decreased. These patients showed favorable outcomes at all myoblast doses, including the highest dose of 600 million cells. On average, OMT patients got worse on each measure.
“These data demonstrate the safety and suggestively positive outcomes of ASM transplantation and warrant initiation of larger Phase 2, double-blind, placebo controlled clinical trials to demonstrate the efficacy of myoblast transplantation in heart failure patients,” Dib said.
Support for this study was provided by Mytogen, Inc.
Statements and conclusions of study authors that are presented at American Heart Association scientific meetings are solely those of the study authors and do not necessarily reflect association policy or position. The American Heart Association makes no representation or warranty as to their accuracy or reliability.
For more information: www.americanheart.org


 November 19, 2021
November 19, 2021