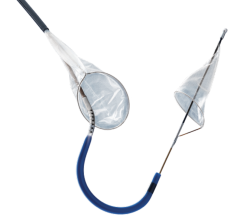
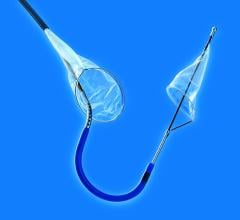
September 9, 2013 — Keystone Heart, a company in the development of cerebral protection devices for interventional cardiology and cardiac surgery procedures, announced its TriGuard cerebral protection device has received CE mark, enabling the company to commercially market the TriGuard in Europe and other territories.
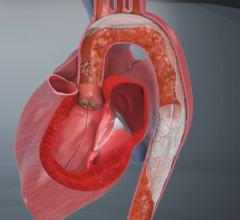
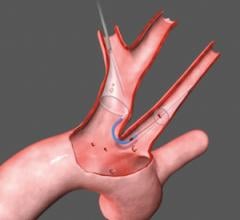
The TriGuard is the only device designed to cover all three aortic cerebral branches to minimize the risk of cerebral damage during transcatheter aortic valve replacement (TAVR) and other cardiovascular procedures. It is the only system designed for placement via one of two femoral artery access ports. The TriGuard accommodates most anatomical variations of the aortic arch and uses a nitinol frame and mesh.
The preservation of cerebral function throughout medical procedures is a key component to procedural success and patient care. Dr. Andreas Baumbach, consultant cardiologist at University Hospitals Bristol, United Kingdom, and honorary reader in cardiology at the University of Bristol, said "The device has the potential to become a routine preventive measure in TAVI and other cardiovascular procedures associated with embolic lesions. European approval of the TriGuard device is an important advancement in patient care."
New scientific data presented at EuroPCR demonstrated a significant reduction of new brain lesion volume during TAVR using TriGuard, compared with historical data on unprotected TAVR procedures. The clinical data shows that maximum total lesion volume was 95 percent smaller and average total lesion volume was 57 percent smaller when compared with historical references.
The medical community is witnessing heightened focus and scrutiny on cerebral injury concurrent with cardiovascular procedures, and research on possible preventative measures and devices to protect against brain injury has increased. Data from the PARTNER A study show nearly twice as many strokes in the TAVR group compared with the surgical aortic valve replacement group at 30 days, the majority of strokes being peri-procedural and within the first 2 days. In addition, newer "next-generation" TAVR devices have so far have apparently failed to reduce TAVR-related risk of stroke. These data affirm that protecting the brain is an important goal during TAVR.
The TriGuard device is not yet commercially available in the United States.
For more information: www.keystoneheart.com


 April 25, 2023
April 25, 2023