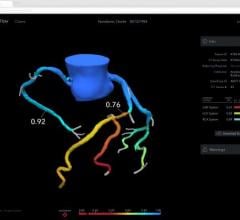
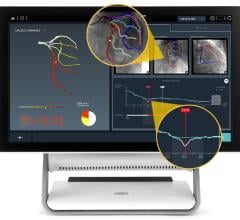
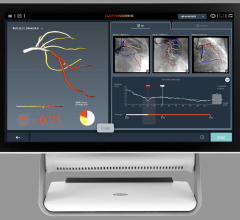
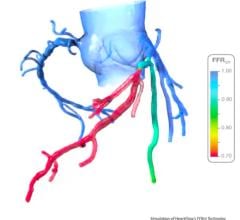
April 2, 2009 - Volcano Corp. this week at ACC 2009 announced the availability of its integrated fractional flow reserve (FFR) technology on all major cardiovascular X-ray systems.
Interest in the use of FFR, a guidewire-based technology that helps assess a lesion’s severity based on the amount of blood flow through the lesion, has increased significantly since the release of the multi-center randomized FAME (FFR vs. Angiography for Multivessel Evaluation) data in late 2008 and publication in the New England Journal of Medicine in January 2009. FAME demonstrated FFR-guidance in routine multi-vessel PCI resulted in clinically-superior outcomes and reduced the composite of death, documented myocardial infarction, and repeat revascularization by 30 percent at one year. The study also demonstrated that these improved clinical outcomes were complemented by significantly reduced cost to the hospitals involved in the trial.
Volcano’s integrated FFR technology can be purchased as part of its s5i system, offering clinicians access to IVUS and FFR at the patient bedside. Volcano has relationships with each of the major cardiovascular X-ray companies to facilitate the installation of the s5i with integrated FFR in any cath lab.
Volcano said it experienced an FFR growth of 36 percent in Q4 2008 versus Q4 2007 and that physicians are increasingly relying on FFR to help guide PCI procedures.
Volcano's integrated s5i was on display at the 2009 ACC meeting in Orlando at the company’s booth as well as in the booths of GE, Philips, Siemens and Toshiba.
For more information: www.volcanocorp.com

 April 30, 2026
April 30, 2026