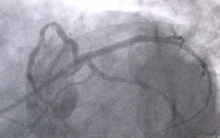
Sudden cardiac death (SCD) is the leading cause of death in high school-aged athletes. To determine if SCD can be prevented with a heart screening, The Christ Hospital Health Network (Cincinnati, Ohio), The University of Mississippi Medical Center and Cincinnati Children’s Hospital Medical Center partnered with USRowing and Toshiba America Medical Systems Inc. to conduct The Athlete Heart Research Study. Initial participants included volunteer high school rowers with more than two years of continuous practice who competed in the USRowing Youth National Championships, June 7 – 9, 2013 in Oak Ridge, Tenn.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now