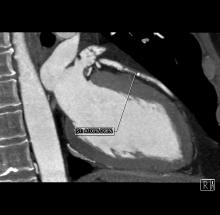
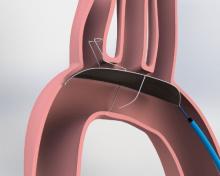
Boston Scientific Corp. has received U.S. Food and Drug Administration (FDA) and CE Mark approval of the Emblem Subcutaneous Implantable Defibrillator (S-ICD) system. The Emblem S-ICD system is a treatment option that provides protection for patients at risk of sudden cardiac arrest (SCA), yet leaves the heart and vasculature untouched, minimizing the risk of complications associated with conventional transvenous implantable cardioverter-defibrillators (TV-ICDs).
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now