As the pioneer of metabolic assessment and behavioral therapy technologies, BodyMedia is developing tools that empower health professionals to better treat patients with chronic health conditions such as obesity, diabetes and cardiovascular disease (CVD).
With BodyMedia’s solutions, clinicians have access to objective health metrics about their patients, which they can use to better educate and inform them about their own bodies and behaviors to help them lose weight and better manage their disease.
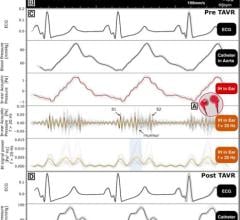
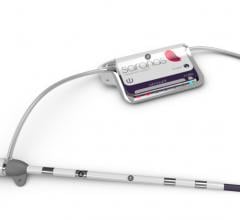
SenseWear Body Monitoring System, allows clinicians to assess their patients using one of the most accurate estimation of energy expenditure today outside of a clinical setting. Worn on the upper arm, the compact, comfortable device has four sensors that track and record complex body statistics such as movement, temperature and heat flux for a variety of clinical applications.
SenseWear Display is the newest enhancement to the Body Monitoring System that captures and displays data transmitted by the SenseWear armband to the SenseWear watchband or the SenseWear clip, which patients attach on their clothing. With the display, patients receive up-to-the-minute reporting of daily calorie expenditure, steps and activity progress.
SenseWear Software allows clinicians to easily upload, analyze and share data recorded by the Body Monitoring System. The information can be used to educate patients on daily metabolic and lifestyle behaviors to improve their health.


 October 21, 2025
October 21, 2025