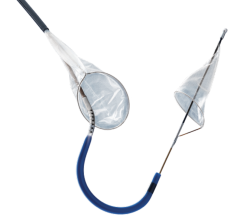
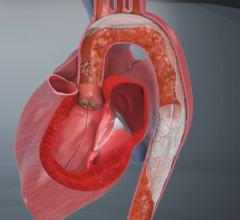
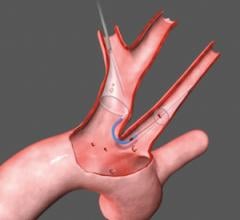
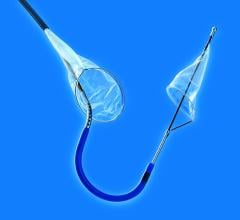
June 1, 2015 — Silk Road Medical Inc. announced the company received U.S. Food & Drug Administration (FDA) 510(k) clearance for its Enroute transcarotid neuroprotection system (NPS). The Enroute transcarotid NPS is used to directly access the common carotid artery and initiate high rate temporary blood flow reversal to protect the brain from stroke while performing carotid angioplasty and stenting (CAS).
"The Enroute technology enables a true hybrid procedure offering the best of both worlds – the critical protection against periprocedural stroke we've achieved with carotid endarterectomy (CEA) with the ability to reduce surgical complications using minimally invasive endovascular techniques (CAS)," said Manish Mehta, M.D., professor of surgery at Albany Medical College and an investigator in the ROADSTER trial. "It is also a quick, efficient procedure which can be performed under local anesthesia with minimal scarring, which is highly beneficial for both the patient and the operator."
Richard Cambria, MD, chief of the Division of Vascular and Endovascular Surgery at Massachusetts General Hospital and the national co-principal investigator of the ROADSTER trial along with colleague Christopher Kwolek, M.D., commented: "We continue to operate on high surgical risk patients because transfemoral CAS has shown excess periprocedural stroke risk. With the Enroute transcarotid NPS, we now have CEA-like neuroprotection and a simplified procedure that can fulfill the promise of CAS."
The FDA cleared the Enroute transcarotid NPS based in part on the results of the ROADSTER trial, which achieved a 30-day stroke rate of 1.4 percent in the pivotal cohort, the lowest to date for any prospective trial of CAS. There were no major strokes and there were no strokes in important high-risk subgroups, including the elderly (age ?75), women and symptomatic patients.
Silk Road Medical has also submitted a premarket approval (PMA) application for the Enroute transcarotid stent system, which is an optimized stent delivery system designed for use with the Enroute transcarotid NPS.
For more information: www.silkroadmed.com


 April 25, 2023
April 25, 2023