Medtronic plc announced that its coronary portfolio will now include the DxTerity Diagnostic…
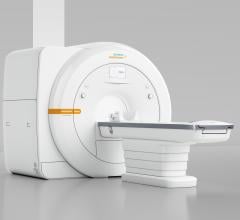
Siemens Healthineers announced that the U.S. Food and Drug Administration (FDA) has cleared the…
Medtronic plc announced the U.S. Food and Drug Administration (FDA) has approved its Freezor…
February 15, 2017 — GE Healthcare announced U.S.
The PRO-Kinetic Energy Cobalt Chromium (CoCr) Coronary Stent System from Biotronik has gained U.…
Biotronik has developed an online tool that streamlines the workflow for physicians selecting…
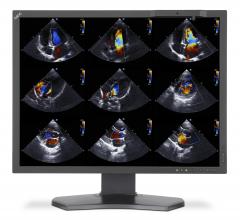
February 13, 2017 — Vital Images Inc.
Vital Images Inc. will feature its Vitrea Modular Enterprise Imaging interoperability solutions…
eHealth Technologies and HealtheConnections, a Syracuse-based regional health information…
Healthcare analytics company Innovaccer Inc. announced the launch of its holistic MIPS Platform…
February 6, 2017 — At the Society of Cardiac Magnetic Resonance (SCMR) 20th Annual Scientific…
Lantheus Medical Imaging Inc. announced U.S. Food and Drug Administration (FDA) approval of a…
Abbott announced U.S. Food and Drug Administration (FDA) approval for magnetic resonance (MR)-…
February 1, 2017 — Medtronic received U.S.
W. L. Gore & Associates (Gore) announced that the Gore Viabahn VBX Balloon Expandable…
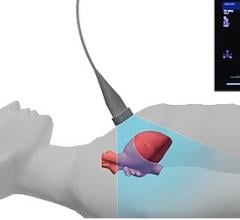
HyperMed Imaging Inc. announced that it has received clearance from the U.S. Food and Drug…
Carestream Health will demonstrate a zero-footprint enterprise imaging platform that can easily…
January 19, 2017 — Apollo Enterprise Imaging Corp.
Intelerad Medical Systems announced a partnership with Laurel Bridge Software that will allow…
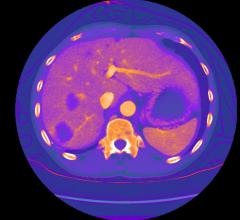
Zetta Medical Technologies announced the release of Zia, an iterative reconstruction solution…

 February 17, 2017
February 17, 2017