Oct. 16, 2025 — Thirona recently announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance ...
Oct. 16, 2025 — Imbria Pharmaceuticals, has announced a partnership with the Hypertrophic Cardiomyopathy Association ...
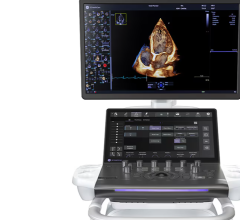
Oct. 15, 2025 — Stereotaxis recently announced it has obtained CE Mark in Europe and submitted a 510(k) application to ...
Sponsored Content — According to the American Heart Association, cardiovascular disease is the leading cause of death in ...
Oct. 15, 2025 — MedAxiom has released its 2025 Cardiovascular Provider Compensation and Production Survey report that ...
Oct. 14, 2025 — Nurea has announced that its PRAEVAorta 2 software has received FDA 510(k) clearance, enabling entry ...
Oct. 13, 2025 — Medtronic plc recently announced it has received U.S. Food and Drug Administration (FDA) labeling ...
Cardiac PET/CT represents a major advancement in cardiovascular diagnostics, offering significant clinical and ...
Oct. 10, 2025 — Johnson & Johnson MedTech, in collaboration with the Heart Rhythm Clinical and Research Solutions, LLC ...
Oct. 10, 2025 — Berlin Heals Holding AG, a clinical-stage medical device company focused on revolutionizing the care of ...
Septe. 30, 2025 -- GE HealthCare Vivid iq Ultrasound system has been selected by NASA for delivery to the International ...
SPONSORED CONTENT — Studycast is a comprehensive imaging workflow system that allows healthcare professionals to work ...
Oct. 8, 2025 — Viz.ai recently launched Viz ACS, a new solution in the Viz Cardio Suite. Designed to unite the acute ...
Oct. 7, 2025 — RSNA Ventures, a mission-aligned subsidiary of Radiological Society of North America (RSNA), has ...
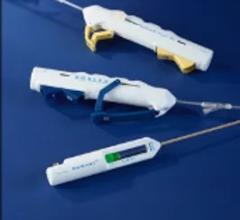
Oct. 7, 2025 — Cagent Vascular has announced the commercial launch of the Serranator PTA Serration Balloon Catheter in 7 ...
Providing exceptional cardiovascular care for patients to achieve the best possible outcomes is the number one goal for ...
Oct. 7, 2025 — Medtronic has announced the full distribution of the Neuroguard IEP System (Neuroguard) after a ...
Oct. 7, 2025 — Nobles Medical Technology II, a provider of cardiovascular closure solutions, has announced that the U.S ...
Oct. 2, 2025 — AorticLab recently announced the U.S. Food and Drug Administration (FDA) has approved its Investigational ...

 October 16, 2025
October 16, 2025