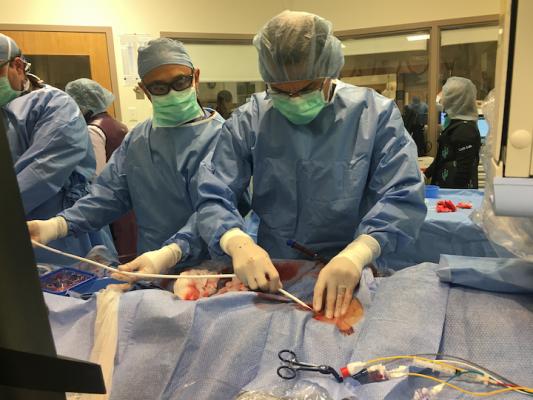
An ECMO cannula being inserted into a cardiovascular patient in need of mechanical oxygenation and hemodynamic support at Tufts Medical Center in December 2019. The FDA just made a temporary approval to use ECMO support in COVID-19 patients during the pandemic emergency. Photo by Dave Fornell.
April 7, 2020 — The U.S. Food and Drug Administration (FDA) has issued guidance to provide a policy to help expand the availability of devices used in extracorporeal membrane oxygenation (ECMO) therapy to address the novel coronavirus (COVID-19, SARS-CoV-2) public health emergency.
ECMO systems are used for long-term respiratory/cardiopulmonary failure and provide assisted extracorporeal circulation and physiologic gas exchange of the patient’s blood for more than six hours. The technology can oxygenate a severely sick COVID-19 pneumonia patient's blood without the need to transfer the oxygen through fluid filled lungs.
As a sudden acute respiratory syndrome, COVID-19 can trigger acute respiratory failure and/or acute cardiopulmonary failure. Under these conditions, the FDA states in the new policy extracorporeal oxygenation can now be used for greater than six hours as tool for treating patients. The FDA said it recognizes the importance and utility of increased availability of ECMO devices for patients during the COVID-19 emergency.
The FDA included a list all the devices that it identified as being technologically capable of being used for ECMO therapy that can now be used for COVID-19 patients.
The FDA also states in the document it does not intend to object to limited modifications to the indications and design of FDA-cleared or FDA-approved ECMO devices and cardiopulmonary bypass devices listed during the declared public health emergency. As long as any modifications do not create an undue risk
As a general best practice, because cardiopulmonary bypass circuit systems might incorporate multiple components, it is recommended that compatibility between components (such as flow rates, coatings, intended patient population) should be considered prior to configuring the circuit for clinical use, the FDA recommends. This includes modifications to cardiopulmonary bypass devices and ECMO devices, such as dimension of cannulae, tubing, filters, connectors, or other accessories to support use in an ECMO circuit that do not affect the flow rate of blood throughout the circuit.
This policy is intended to remain in effect only for the duration of the public health emergency related to COVID-19 declared by the Department of Health and Human Services (HHS). This period will include any renewals made by the HHS secretary.
The FDA included notice of this policy chang in the document "Enforcement Policy for Extracorporeal Membrane Oxygenation and Cardiopulmonary Bypass Devices During the Coronavirus Disease 2019 (COVID-19) Public Health Emergency."
Download the complete ECMO document from the FDA.
Related ECMO Use in COVID-19 Content:
FDA Approves ECMO to Treat COVID-19 Patients
VIDEO: ECMO Support Effective in Sickest COVID-19 Patients — Interview with Ryan Barbaro, M.D.
ECMO Used to Treat Adult Respiratory Distress Syndrome Case
ECMO Support Found Effective in Sickest of COVID-19 Patients
LivaNova Modifies its ECMO Indications Beyond Six Hours to Address COVID-19
VIDEO: Multiple Cardiovascular Presentations of COVID-19 in New York — Interview with Justin Fried, M.D., explaining a case that used VV-ECMO abnd VAV-ECMO
New York City Physicians Note Multiple Cardiovascular Presentations of COVID-19
Related Cardiology Related COVID-19 Content:
VIDEO: Cancelling Non-essential Cardiac Procedures During the COVID-19 Outbreak — Interview with Ehtisham Mahmud, M.D.
ACC COVID-19 recommendations for the cardiovascular care team
VIDEO: What Cardiologists Need to Know about COVID-19 — Interview with Thomas Maddox, M.D.
The Cardiac Implications of Novel Coronavirus
ESC Council on Hypertension Says ACE-I and ARBs Do Not Increase COVID-19 Mortality
VIDEO: Imaging COVID-19 With Point-of-Care Ultrasound (POCUS)
CT Provides Best Diagnosis for Novel Coronavirus (COVID-19)
Radiology Lessons for Coronavirus From the SARS and MERS Epidemics
Deployment of Health IT in China’s Fight Against the COVID-19 Epidemic
Radiologists Describe Coronavirus CT Imaging Features
Coronavirus Update from the FDA
CT Imaging of the 2019 Novel Coronavirus (2019-nCoV) Pneumonia
Additional COVID-19 Resources for Clinicians:
Johns Hopkins Coronavirus Resource Center with inteavtive map of cases in U.S. and worldwide
World Health Organization (WHO) COVID-19 situation reports
World Health Organization (WHO) coronavirus information page
U.S. Food and Drug Administration (FDA) COVID-19 information page
Centers for Disease Control (CDC) COVID-19 information page


 May 05, 2026
May 05, 2026 









