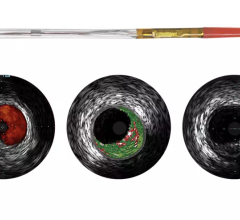
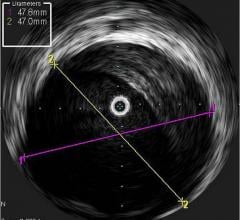
An example of OCT showing a stent with neointimal growth over the stent struts. The struts appear as bright spots.
Imaging technology and diagnostic tools for interventional cardiologists have changed dramatically over the past few years with the introduction of optical coherence tomography (OCT), improvements in intravascular ultrasound (IVUS) and pressure guide wires used in fractional flow reserve (FFR). Product innovation and integration of these technologies, as well as a growing base of clinical evidence, are transforming the way physicians diagnose, treat and follow-up patients undergoing percutaneous coronary intervention (PCI).
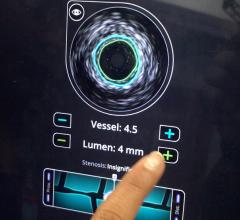
IVUS allows physicians to visualize lumen diameter and vessel walls from within the vessel, providing better guidance for stent placement. Although IVUS came to market more than two decades ago, use in the United States is estimated at approximately 15 percent of PCI procedures, with image quality, time and cost (due to lack of reimbursement) cited as limiting factors to adoption. In Japan however, IVUS has reimbursement and is used in more than 70 percent of PCI procedures.
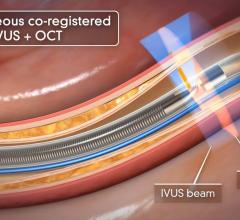
While IVUS provided better information than angiography alone, poor image quality led manufacturers to begin researching other imaging technologies. OCT was brought to market first in Japan by LightLab Imaging in 2008, and launched in the United States in May 2010. OCT uses near infrared light to project images of the vessel wall and lumen. Due to the high frequency associated with the light signal, image resolution is 10 times greater than that of IVUS, leading to better views of stent apposition and thrombus detection. Reduction in stent mal-apposition, which is associated with late stent thrombosis (LST) and better resolution for neo-intimal tissue growth and in-stent restenosis are some of the key advantages of OCT.
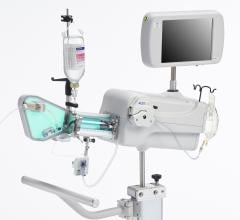
St. Jude Medical purchased Lightlab Imaging in May 2010, and its C7-XR intravascular imaging system and C7 Dragonfly catheter have been available in the United States for about a year, but have yet to make waves. An integrated FFR/OCT platform is expected to launch in the United States during the second half of 2011. The integrated platform will have wireless capability, allowing for data and images to be sent to lab recording stations, without the trouble of moving consoles between labs. The importance of FFR – which the European Society of Cardiology (ESC) recently promoted to the highest level of clinical evidence – and its use through an integrated OCT platform should increase the awareness and use of OCT in the medium term.
Volcano Corp. is developing a multimodality IVUS/OCT/FFR system that is expected to be a complete and convenient diagnostic toolbox for interventional cardiologists. The integrated platform will provide better access to all three technologies, save time switching between consoles and catheters and could theoretically represent cost savings in capital expenditure as well as disposables. The company will begin clinical trials for its product in the United States and Japan this year. It also is expected to gain CE mark and launch in Europe during the second half of 2011, with a launch in the United States and Japan planned for 2012 and 2013, respectively. The Volcano OCT system will boast faster pullback, made possible through a smaller catheter. A faster pullback will mean less contrast media will be required and hence reduced side effects for patients.
Evidence Angiography Alone is Insufficient
Recently, both St. Jude Medical and Volcano acknowledged that OCT is used primarily as a clinical research tool. They also said physicians currently struggle with when to use it, what it measures, how to interpret results and how to use it with angiography, IVUS or FFR.
In recent years, significant clinical evidence has been published to demonstrate that suboptimal PCI is frequent and impacts long-term efficacy and safety. The STLLR trial showed that geographic miss (GM) occurred in as many as 66 percent of patients, and that GM leads to higher risk of target vessel revascularization (TVR) and myocardial infarction (MI) at one year. Similarly, a recent article in The New England Journal of Medicine reported that angiography is only correct in 54 percent of procedures, creating further support and interest in using new imaging technologies that provide a three-dimensional view of the vessel and can measure plaque severity and vessel tissue characterization.
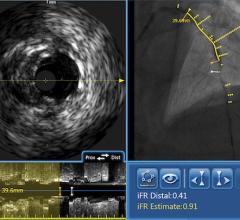
Several studies have been published in support of IVUS-guided interventions, in particular for stenting in the left-main coronary artery, bifurcated lesions, and the superficial femoral artery. The MAIN-COMPARE trial demonstrated that IVUS-guided patients had lower mortality at three years. Support for IVUS and virtual histology (VH)-IVUS was published in the PROSPECT trial, which showed that angiography was not a good predictor of future events and that IVUS and VH-IVUS can predict lesion risk for future cardiac events. FAME showed an unequivocal benefit in using FFR for lesion assessment.
The case to use more than angiography in the diagnosis and treatment of coronary artery disease is now well established, but manufacturers need to establish the role and evidence in support of OCT. Both St. Jude and Volcano have trials planned for this year, and as their integrated platforms are launched, we can expect to learn more about the benefits of using OCT.
Editor’s note: Millennium Research Group (www.MRG.net), a Decision Resources Inc. company (www.DecisionResourcesInc.com), is an authority on medical technology market intelligence. The company provides specialized industry expertise through syndicated reports, ongoing Marketrack projects, customer loyalty tracking, facility-level procedure forecasting and customized solutions.




 September 18, 2025
September 18, 2025