
An example of intra-cardiac echo (ICE) imaging from Siemens Healthineers showing color flow Doppler with severe valve regurgitation. ICE has seen wider use in transcatheter structural heart procedures because, unlike TEE, it can be used without deeper sedation of anesthesia.
Intra-cardiac Echocardiography (ICE) uses catheter-based cardiac ultrasound array to image anatomy and devices inside the heart to help guide procedures. ICE is primarily used in electrophysiology (EP) lab ablation procedures and transcatheter structural heart interventions.
Both transthoracic (TTE) and transesophageal echocardiography (TEE) are used for guiding transcatheter interventions, but they have limitations. ICE provides the advantage of imaging from within the heart, providing shorter image distances and higher resolution. ICE may be performed without sedation and avoids esophageal intubation as with TEE. But, limitations of ICE include the need for additional venous access with possibility of vascular complications, potentially higher costs and a learning curve for new operators.[1]
ICE can be helpful in guiding transseptal punctures for both EP lab and structural heart device transseptal punctures (including left atrial appendage occlusions), transcatheter aortic valve replacement (TAVR), transcatheter mitral valve repair (TMVR), and transcatheter closure of paravalvular leak (PVL), patent ductus arteriosus (PDA), atrial septal defects (ASD), ventricular septal defect (VSD), and patent foramen ovale (PFO).[2]
ICE has been widely adopted in the EP lab to guide transseptal punctures due to its ability to define atrial septal anatomy and provide visualization of transseptal catheter position in relation to other structures within the heart.[3]
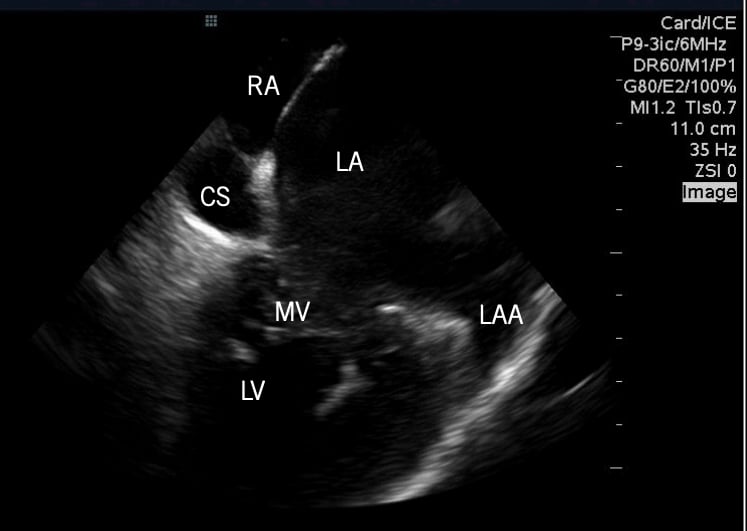
Example of the Abbott/St. Jude Medical 2-D intra-cardiac echo (ICE) system showing cardiac anatomy.
Advantages and Disadvantages of ICE vs. TEE for Structural Heart Procedural Imaging
Intracardiac echo has the advantage of being able to image structures near the right atrium, it only requires minimal sedation and it can be performed by interventionalist.[2] However, ICE has limited 3-D evaluation, limited temporal resolution, it carries a risk of vascular access complications and it has a high upfront cost.
Transesophageal echo offers a more comprehensive examination, the ability to perform a full 3-D evaluation and has improved temporal resolution. The downsides of TEE include the need for sedation or anesthesia, the probe interfers with fluoroscopic imaging and it requires and echocardiographer to perform the exam. There also is a risk of trauma to the gastrointestinal tract, which could result in an increased length of stay.[2,4]
ICE Moves to 3-D and 4-D Imaging
In the past decade, ICE has moved from 2-D echo to 3-D and 4-D echo imaging capabilities. This has greatly enhanced device guidance visualization. The 2-D ICE systems enabled bi- or tri-plane imaging, showing two or three different imaging plane views, but the sonographer and the operator need to reconstruct these images into what the 3-D anatomy and device alignment looked like in their heads. For operators unfamiliar with sonography, it was often difficult to understand what they were looking at. 3-D imaging and 4-D imaging (dynamic 3-D in motion during live imaging) now enables easier interpretation of the images from a more realistic surgeon's view inside the heart. This is especially helpful in structural heart interventions.[5]
This 3-D ultrasound data is captured in a volume. To explain this for non-sonographers, picture a 3-D cube in your mind, as opposed to a 2-D slice that is more like looking as piece of paper, or one thin slice of that 3-D cube. As computing power and processing speeds increase, ultrasound systems have been able to process these larger volumes of data to quickly reconstruct the images in real-time. Faster computers also have enabled faster frame rates so the resulting dynamic, live imaging appears more life like and less jerky.
This type of 3-D/4-D capability is available on the Siemens Healthineers Acuson SC2000 Prime ultrasound system.
Forward Looking ICE
In 2017, Conavi received FDA clearance for an enhanced version of its Foresight ICE system, which is unique in that it allows forwarding looking ICE, rather than imaging from the side of the catheter. The system also enables color Doppler, pulsed wave Doppler, 2-D and 3-D measurements and electrocardiogram (ECG)-gated 3-D image acquisition. It offers an 8 cm radius field-of-view with high near-field resolution. Imaging is possible within 3 mm of the catheter tip. Combined with an atraumatic tip, users can position the Foresight ICE catheter adjacent to structures and areas of interest. Since the imaging plane is perpendicular to the shaft of the catheter, it is more intuitive to interpret the image and understand the orientation in the anatomy.
Integration of ICE and IVUS Into One System
Boston Scientific has a point of difference with its iLab Polaris system in that it supports both ICE and intravascular ultrasound (IVUS) used in coronary and peripheral artery interventions to visualize atherosclerotic lesions and stent apposition in the cath lab. The IVUS system also has lumen border detection and plaque characterization software to help define areas of calcified, fibrotic or lipid core plaques to help guide revascularization procedures.
ICE Systems from Abbott, Philips, GE and Biosense Webster
Abbott Vascular, through an acquisition of St. Jude Medical, offers the ViewFlex Xtra ICE Catheter, which is compatible with the ViewMate Z and ViewMate II ultrasound consoles. Philips is the original equipment manufacturer (OEM). The ICE catheters can also be used with the Philips CX50 xMatrix ultrasound system. The system provides visualization of cardiac structures and blood flow within the heart during electrophysiology and interventional cardiology procedures. Here is a link to a helpful, illustrated user manual for ICE imaging with this system.
In 2013, through its partnership with St. Jude, Philips introduced the CX50 xMatrix, which was the first portable ultrasound at the time with live 3-D TEE and 2-D ICE capability.
GE Healthcare also offers portable ultrasound systems with ICE imaging capability on its Vivid S70N v204, Vivid iq 4D v204 and Vivid iq Premium v204 ultrasound systems. However, GE does not make its own ICE imaging catheters. The GE systems are compatible with the Biosense Webster CartoSound ICE Module.
The Biosense Webster CartoSound is a module of the widely used Carto 3 EP electromapping system used in cardiac ablations. The system can use its own brand of CartoSound ICE catheters, or the Siemens Acuson AcuNav ICE catheter. The system delivers real-time ICE imaging and navigation. The use of ICE also can help reduce the use of fluoroscopy and extra radiation dose during these long EP procedures.
Related Intra-cardiac Echo Content:
Northwestern Medicine First in U.S. to Use Live 3D Intracardiac Echo for EP Procedure
Mayo Clinic First to Use Next Generation 4D ICE System to Guide Structural Heart Procedures
Trends and Advancements in Intra-cardiac Echo Imaging Systems
Intra-Cardiac Echo Aids EP Ablations, Structural Heart Procedures
Intracardiac Echo Should be More Widely Adopted to Minimize Radiation in Catheter Ablations
References:


 May 13, 2026
May 13, 2026 









