September 25, 2019 — Ten-year survival rates are similar for bypass surgery and coronary stenting with drug-eluting ...
Cardiovascular Surgery
This channel includes news and new technology innovations for cardiac surgery. This includes coronary artery bypass grafts (CABG) and surgical valve replacements.
August 22, 2019 — The amount of opioids prescribed for patients after heart and lung surgery has a direct relationship ...
August 1, 2019 — Less-invasive procedures to open severely clogged leg arteries were as good at helping people survive ...
July 26, 2019 — A highly competitive $4.5 million grant from the National Institutes of Health (NIH) National Heart ...
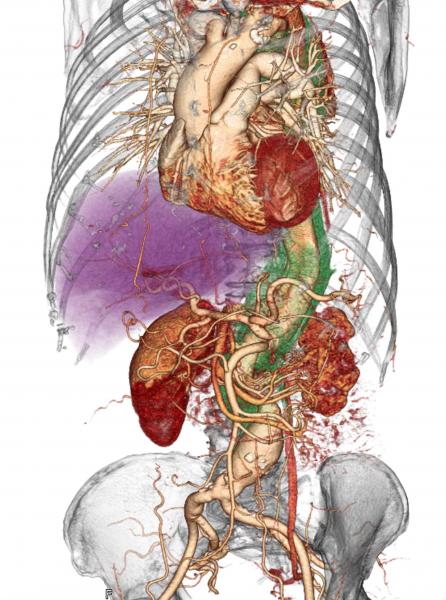
July 18, 2019 — When a patient comes to the hospital with acute type A aortic dissection (ATAAD), a tearing of the inner ...
July 12, 2019 — Internet search engine giant Google unveiled a new Doodle on its homepage Friday, July 12, 2019 ...
David M. Axelrod, M.D., clinical associate professor in pediatrics (cardiology) at Stanford School of Medicine, explains ...
May 13, 2019 — A randomized clinical trial effectively used nerve stimulation through an ear clip to reduce atrial ...
May 3, 2019 — Coronary artery bypass grafting (CABG) surgery may be the best treatment option for most patients with ...
April 19, 2019 — Aziyo Biologics Inc. announced the publication of results from its landmark RECON study in the Journal ...
Here is what I thought were the top five most important take-away presentations from the 2019 Society of Thoracic ...
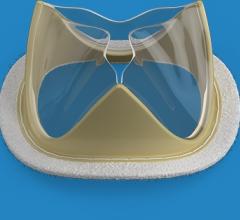
February 18, 2019 — Foldax Inc. announced the U.S. Food and Drug Administration (FDA) has granted investigative device ...
February 14, 2019 — Medical imaging and visualization company Medivis officially unveiled SurgicalAR, its augmented ...
February 1, 2019 — Paragonix Technologies Inc. recently received clearance from the U.S. Food and Drug Administration ...
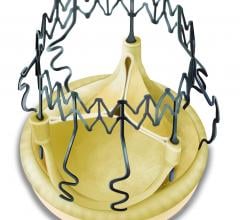
January 29, 2019 — LivaNova PLC announced the publication of three separate studies highlighting the performance of its ...

 September 25, 2019
September 25, 2019