April 5, 2011 – A microcatheter system has received the CE mark. The Plato Microcath system, by Scientia Vascular ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).

April 5, 2011 – Death rates are similar at one year for a catheter-based aortic valve replacement procedure and ...
March 29, 2011 – The U.S. Food and Drug Administration (FDA) cleared a wireless point-of-care testing system that ...
Venous thromboembolism (VTE), and pulmonary embolism (PE) in particular, are among the most complex and dangerous acute ...
March 28, 2011 – Patients who have suffered a "mini stroke" are at twice the risk of heart attack than the ...
March 25, 2011 – The American College of Cardiology (ACC) and the American Heart Association (AHA) have released a ...
Radial access is very popular outside the United States because it offers improved safety, comfort and cost savings over ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
St. Joseph's Hospital of Atlanta created a radial access recover room that replaces beds and the sterile institutional ...
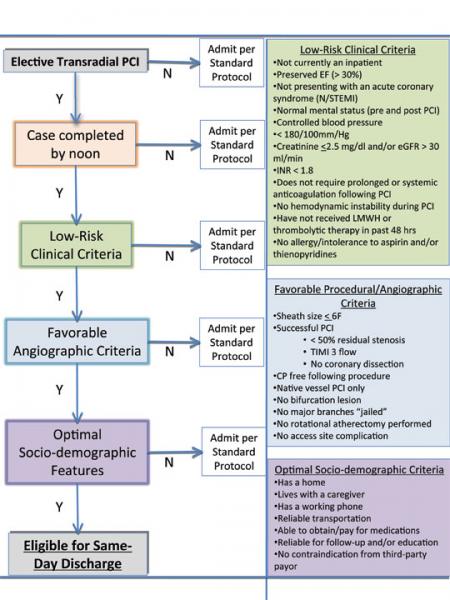
With the increased use of transradial cardiac catheterization throughout hospitals across the country, several results ...

Heart disease remains the number one killer of both men and women in the United States, with ST elevation ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...

Over the last 10 years, hospitals have phased out analog image intensifiers for flat-panel, digital detector angiography ...
March 18, 2011 – A clinical trial testing a robotic system to remotely deliver and manipulating coronary guide ...
March 18, 2011 – Circulating through the bloodstream of every human being is a rare and powerful type of cell, one ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
March 10, 2011 – A newly introduced catheter provides a superior answer for percutaneous fluid aspirations and ...
Physio-Control has acquired Swedish-based Jolife AB, the developer and manufacturer the automated Lucas Chest ...
Circadian rhythms affect the incidence of coronary stent thrombosis, with more events occurring during the early ...

 April 05, 2011
April 05, 2011