July 31, 2009 – A United Kingdom court has determined Occlutech GmbH’s products do not infringe on AGA Medical ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
July 30, 2009 – A new study comparing nephrotoxicity of the iso-osmolar contrast medium iodixanol to low-osmolar ...
July 29, 2009 – Medtronic Inc. said earlier this week it has reached a settlement with Abbott Laboratories over ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
July 28, 2009 – Onset Medical Corporation said today it received CE mark approval to begin marketing the SoloPath ...
July 27, 2009 – Prescient Medical Inc. received the prestigious 2009 North American Product Innovation of the ...
July 23, 2009 – The FDA recently cleared Ziehm Imaging’s latest mobile C-arm, the Ziehm Vision RFD, which is ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
July 22, 2009 – W. L. Gore & Associates today said it received FDA approval to market its GORE VIABAHN ...
July 21, 2009 – Total procedures performed in angio labs from 2004 to 2008 grew 20 percent, from 4 million in 2004 ...
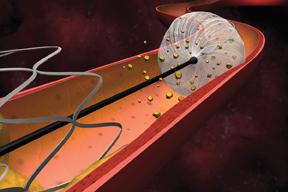
When stenting emerged as a minimally- invasive treatment for clogged arteries associated with peripheral artery ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
In the current economy where hospitals are tightening their belts to help get through tough times, technology can ...

The use of remote, prehospital ECG transmission systems by emergency medical services (EMS) can help quickly ...

Interventionalists using radial artery access instead of femoral access say it significantly cuts bleeding complications ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
July 17, 2009 – Medtronic announced today the completion of a 12-month follow-up in the STOP-AF (Sustained ...
July 16, 2009 – Cook Medical received clearance today from the FDA to market its newest balloon dilatation ...
July 16, 2009 – Boston Scientific Corp. today said its received approval from the FDA to market its TAXUS Liberte ...

 July 31, 2009
July 31, 2009