September 18, 2009 ¬– Abbott has announced the company's schedule of key news announcements and events during the ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
September 17, 2009 – NMT Medical Inc. said yesterday it will commence data analysis for its landmark STARFlex ...
September 17, 2009 – Boston Scientific Corp. announced its schedule of the company's major events and press ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
September 17, 2009 – Elixir Medical Corp. said today its stent systems using bioabsorbable polymers will be ...

In August Daiichi Sankyo and Eli Lilly and Company launched U.S. sales of Effient (prasugrel), a new antiplatelet ...
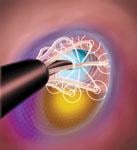
Ruptured vulnerable plaque (VP) is blamed for the majority of acute myocardial infarctions and many strokes, but ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...

Stenting of lesions at coronary vessel bifurcations is one of the most challenging percutaneous interventions ...

Over the past year several new drug-eluting stents (DES) stents have been released and SYNTAX trial data proved ...

Metal stents are foreign objects that can irritate vessels and cause stent thrombosis, prevent vessel ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...

The polymer coating on drug-eluting stents (DES) used to carry the antiproliferative drugs cause an increased risk ...
September 16, 2009 – Texas Cardiac Arrhythmia Institute at St. David’s Medical Center in Austin, Texas plans to ...
September 16, 2009 – Hansen Medical Inc. today said it received FDA clearance for its next generation Sensei X ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
September 15, 2009 – Tryton Medical Inc. said today its Tryton Side Branch Stent has been used in 250 procedures ...
September 14, 2009 – OrbusNeich's Genous Bio-engineered R stent is feasible and safe in patients who need ...
Sept. 14, 2009 – Abbott said today the Chinese State Food and Drug Administration (SFDA) has approved the XIENCE ...


 September 17, 2009
September 17, 2009