February 24, 2014 — TeraRecon, a provider of medical image management solutions, debuted it’s iNteract+ solution at the ...
Diagnostic and Interventional Cardiology DAIC
Diagnostic and Interventional Cardiology (DAIC) is a magazine that reaches more than 25,000 healthcare professionals in cardiology, interventional cardiology and cath labs across the United States. These influential buying team members rely on DAIC's award-winning editorial content and comparison charts as a unique research tool for specifying, recommending and approving technology/device purchases.
February 21, 2014 — ScImage announced availability of a Master Patient Index (MPI) translator that provides healthcare ...
February 20, 2014 — Healthcare expenditure has been increasing throughout the last decade, and is beginning to become ...
February 20, 2014 — Blackford Analysis, a software provider with products to accelerate medical image comparison ...

February 19, 2014 — A new treatment option for heart disease is being tested at The University of Kansas Hospital that ...
February 18, 2014 — Neovasc Inc. announced that a first-in-human implantation of its Tiara transcatheter mitral valve wa ...
February 18, 2014 — HIT Application Solutions announced a partnership with Merge Healthcare Inc. to integrate its Notifi ...
February 18, 2014 — The U.S. Food and Drug Administration (FDA) cleared Carestream Health’s Vue Motion image viewer for ...
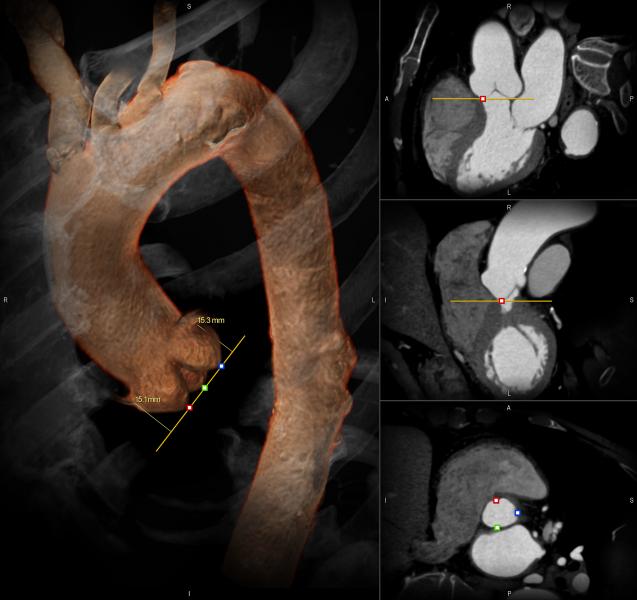



 February 24, 2014
February 24, 2014