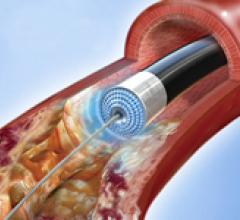
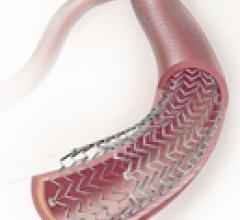
October 1, 2009 – Flexible Stenting Solutions Inc. (FSS) said this week it received conditional investigational ...
Peripheral Artery Disease (PAD)
This channel includes news, interventions, and new technology innovations for peripheral artery diease, PAD and critical limb ischemia.
October 1, 2009 – Invatec recently launched the REEF HP PTA Balloon Catheter at CIRSE 2009, which the company said ...
October 1, 2009 – Spectranetics Corp. yesterday said it filed a 510(k) application with the U.S. Food and Drug ...
September 30, 2009 – Invatec this week at the European CIRSE 2009 conference launched its latest self-expanding ...
September 21, 2009 — Minnow Medical recently announced it will be presenting interim clinical data regarding a ...

Bare metal stents (BMS) were introduced in 1994 as an improvement over balloon angioplasty alone, which had a high ...
September 11, 2009 – Terumo Interventional Systems yesterday launched its Glidewire Advantage Peripheral Guidewire ...
The HI-TORQUE Versacore .035 Guide Wire is a peripheral guide wire is designed for routine diagnostic and device ...
The FoxCross PTA Catheter is a next-generation .035 balloon dilatation catheter is used to open peripheral ...
August 31, 2009 – IDEV Technologies Inc. announced last week the first patient was enrolled in the company's FDA ...
August 11, 2009 – Pathway Medical Technologies Inc. today introduced the 7 French compatible version of its ...
August 11, 2009 – Physicians in the United Kingdom yesterday completed patient implants using the first CE mark ...
July 22, 2009 – W. L. Gore & Associates today said it received FDA approval to market its GORE VIABAHN ...
July 17, 2009 – AGA Medical Corp. received European CE mark approval for its AMPLATZER vascular plug, AVP 4, which ...
July 16, 2009 – Cook Medical received clearance today from the FDA to market its newest balloon dilatation ...

 October 01, 2009
October 01, 2009