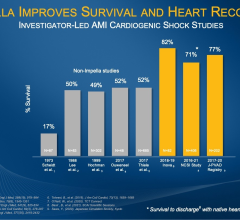
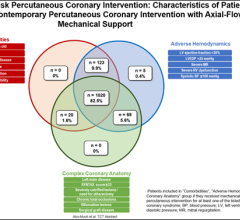
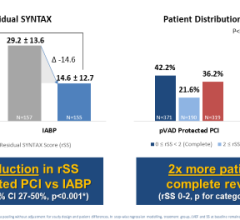
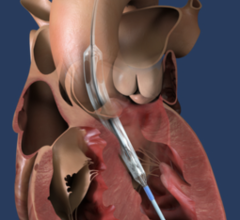
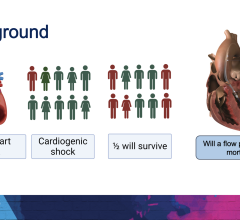
Abiomed (NASDAQ: ABMD) is a pioneer and global leader in healthcare technology and innovation, focused on breaking new ground in recovering hearts and saving lives. Because hundreds of people die each year waiting for heart transplants, Abiomed CEO, Chairman and President, Michael R. Minogue has focused the company’s efforts on developing ground-breaking “bridge to recovery” medical technologies designed to assist or replace the life-sustaining pumping function of the failing heart. The company’s portfolio of heart recovery products and services offer healthcare professionals an array of choices across a broad clinical spectrum. Abiomed’s minimally invasive devices enable the heart to rest, heal and recover by improving blood flow and circulatory support. Through the use of Abiomed’s heart recovery products, patients can return home to live longer and fuller lives with their own native heart. Abiomed was founded more than two decades ago in 1981 and is the only company in the world with exclusive labeling on circulatory support and ventricular assist devices (VADs) for all potentially recoverable heart failure indications. As the global leader for products in the acute heart failure market, the Company ships more heart recovery pumps and VADs than any other company in the world. With a growing staff of over 300 employees, Abiomed is headquartered in Danvers, Mass., and additional offices in Aachen, Germany and Ireland. From the world’s first total replacement heart to a catheter-based device that provides circulatory support with rapid and simple insertion, Abiomed is dedicated to finding ways to treat heart failure so that patients and physicians have the most advanced, successful, and beneficial technology available to improve quality of life.
22 Cherry Hill Drive
Danvers , MA 01923
USA
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now

 April 08, 2024
April 08, 2024