Getty Images
January 25, 2024 — Agepha Pharma, a leading multinational pharmaceutical company with the first FDA-approved anti-inflammatory cardiovascular treatment (LODOCO– colchicine, 0.5 mg tablets), together with Caristo Diagnostics, a leading cardiac disease diagnostics company with the novel CaRi-Heart technology to visualize and quantify coronary inflammation, announced today their collaboration to improve awareness and clinical education about the central role of inflammation in coronary artery disease.
The companies will jointly support a series of educational forums among clinicians beginning later this year highlighting the significant new scientific breakthroughs that are radically transforming the traditional approach to heart disease prevention and prediction. They will also selectively support clinical research involving the CaRi-Heart technology for coronary inflammation diagnostics and LODOCO 0.5 mg tablets for therapeutic intervention.
“Caristo shares Agepha Pharma’s passion for fighting coronary inflammation, which is one of the primary drivers of heart attack risks,” said Frank Cheng, CEO of Caristo Diagnostics. “We look forward to collaborating with Agepha Pharma in order to combat heart disease as the number one cause of death globally.”
“Together with Caristo we are proud to lead this revolution in preventative cardiology,” said Steve Andrzejewski, Agepha Pharma Head of US Operations. “In the future, patients with coronary inflammation can be identified by the CaRi-Heart technology and treated by our FDA-approved LODOCO 0.5 mg tablets.”
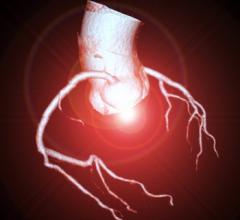
More than 8 million Americans visit hospital emergency departments experiencing chest pain every year. Coronary computed tomography angiography (CCTA) is routinely used to diagnose CAD caused by the narrowing (stenosis) or blockage of the coronary arteries that supply the heart with blood. But in three quarters of patients, there is no clear sign of significant narrowing at the time of CCTA. Nonetheless, many of these people still die from a heart attack within a few years, driven by inflammation that is now detectable in routine CCTA scans with the CaRi-Heart technology. Clinical trials have shown significant reductions in cardiovascular risk through therapeutic targeting of coronary inflammation with anti-inflammatory treatments such as colchicine, but identifying those at highest risk due to undiagnosed coronary artery inflammation has great potential for more patients to be treated effectively, to prevent more heart attacks and save more lives.
Agepha’s LODOCO inhibits microtubule assembly and has multiple anti-inflammatory mechanisms. In June 2023, the U.S. Food and Drug Administration (FDA) approved LODOCO as the first anti-inflammatory atheroprotective cardiovascular treatment demonstrated to reduce the risk of myocardial infarction (MI), stroke, coronary revascularization, and cardiovascular death in adult patients with established atherosclerotic disease or with multiple risk factors for cardiovascular disease. The effectiveness and safety of LODOCO in preventing heart attacks and strokes is supported by randomized trial data reported in the New England Journal of Medicine, Circulation, Journal of the American College of Cardiology, and European Heart Journal, while data emphasizing the critical need to address inflammation as much as high cholesterol in heart disease patients has recently been described in The Lancet. Specifically, LODOCO reduces cardiac event risk in adult patients with established atherosclerotic cardiovascular disease (ASCVD) by an additional 31% as compared to placebo.
Caristo’s CaRi-Heart technology applies advanced AI algorithms to routine cardiac CT scans to visualize and quantify coronary inflammation. Since 2018, exceptional validation results showing CaRi-Heart technology’s ability to aid the prediction of heart attacks have been published in leading medical journals including the Lancet, JACC, European Heart Journal, and Cardiovascular Research. New research recently presented at the American Heart Association’s Late Breaking Science Sessions has found that the CaRi-Heart technology can predict fatal and non-fatal cardiac events independently from routine clinical risk scores and standard coronary computed tomography angiography (CCTA) interpretation, and can significantly impact treatment for CAD by transforming risk stratification and management of patients undergoing routine CCTA. Among patients who had no plaque and zero calcium at time of CCTA, those with the most abnormal CaRi-Heart test results experienced 11-fold higher risk for cardiac mortality and 5-fold higher risk for major adverse cardiac events (MACE) than those who had normal CaRi-Heart test results. The CaRi-Heart technology is in clinical use in the UK, EU and Australia, and is limited to research use in the U.S. before the FDA grants marketing authorization.
For more information: https://us.agephapharma.com/
Related content:
U.S. FDA Approves First Anti-Inflammatory Drug for Cardiovascular Disease
Lowering Atherosclerotic Cardiovascular Disease Events by Treating Residual Inflammatory Risk
U.S. FDA Approves First Anti-Inflammatory Drug for Cardiovascular Disease

 February 06, 2026
February 06, 2026