February 3, 2017 — Avinger Inc. recently announced positive two-year clinical data from the pivotal VISION study of the company’s Lumivascular technology. The VISION study was designed to evaluate the safety and effectiveness of Avinger’s Pantheris system to perform directional atherectomy while, for the first time ever, allowing physicians to use real-time intravascular imaging to aid in the removal of plaque from diseased lower extremity arteries. Data from the study, which demonstrated successful achievement of all primary and secondary safety and effectiveness endpoints, supported U.S. Food and Drug Administration (FDA) 510(k) clearance of the system in 2016.
Interim results from 55 patients who completed 24-month follow-up were presented by Ian M. Cawich, M.D., cardiologist at Arkansas Heart Hospital, at the Leipzig Interventional Course (LINC) in Leipzig, Germany.
Key highlights of the presentation include:
- Kaplan Meier survival probability measured 86 percent freedom from target lesion revascularization (TLR) at 12 months and 82 percent freedom from TLR at 24 months;
- Zero (0 percent) amputations through 24 months;
- Statistically significant improvements in ankle brachial index (ABI) and Rutherford classification at 30 days maintained through 24 months; and
- A majority of lesions were treated using standalone optical coherence tomography (OCT)-guided atherectomy with the Pantheris catheter, with only 9.6 percent (n=19/198) receiving adjunctive drug coated balloon (DCB) therapy and only 5 percent (n=10/198) receiving adjunctive stent therapy.
“These interim 24-month results demonstrate a stable, consistent, and high-functioning patient population through two years – impressive results for a study of mostly standalone, drug-free atherectomy cases,” said Cawich. “The findings are particularly striking considering that all data generation and outcomes were in the setting of a first-generation clinical trial device and brand new users.”
In addition, Marianne Brodmann, M.D., of the Medical University of Graz in Austria, presented a prospective case series of 30 patients (35 lesions) with long-term follow up including duplex patency and freedom from TLR at six and 12 months.
The study represented the first cohort of Pantheris outcomes patency data (PSVR<2.4), with a rate of 89 percent at six months and 86 percent at 12 months. OCT-guided atherectomy demonstrated 84 percent standalone luminal gain, and OCT-guided CTO crossing achieved 95 percent (n=20/21) standalone true lumen crossing. At six months, freedom from TLR of 94.3 percent closely matched VISION six-month results of 94 percent; freedom from TLR at 12 months of 88 percent also approximates VISION 12-month results of 86 percent. In addition, the study demonstrated a statistically significant improvement in ABI from baseline to both six and 12 months.
“As a new user, I am very encouraged by our positive long-term results,” said Brodmann. “Lumivascular treatment demonstrated tremendous efficacy and safety, consistent with the larger pivotal VISION study.”
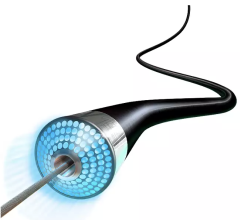
Atherectomy is a minimally invasive treatment for peripheral artery disease (PAD) in which a catheter-based device is used to remove plaque from a blood vessel. Lumivascular technology allows physicians to see from inside the artery during a directional atherectomy procedure by using OCT, which is displayed on the Lightbox console. In the past, physicians have had to rely solely on X-ray as well as tactile feedback to guide their tools while treating complicated arterial disease. With the Lumivascular approach, physicians can more accurately navigate their devices and treat PAD lesions, thanks to the real-time OCT images generated from inside the artery, without exposing healthcare workers and patients to the negative effects of ionizing radiation.
For more information: www.avinger.com


 November 08, 2024
November 08, 2024