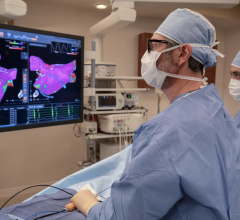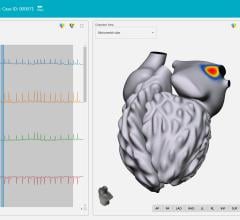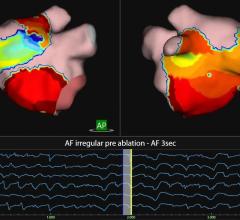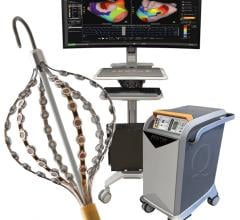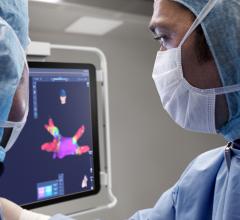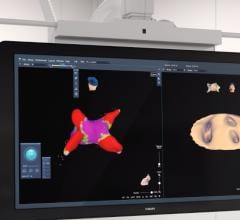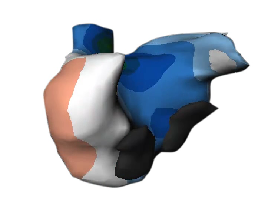
The ECVUE System provides detailed images of the heart
February 24, 2012 — CardioInsight Technologies Inc. announced that it has successfully completed the European commercial introduction of its ECVUE system to aid in the diagnosis of cardiac arrhythmias. St. Mary's Hospital in London became the company's first commercial customer. Doctors Prapa Kanagaratnam, Wyn Davies, Nick Peters, David Lefroy, Boon Lim and Shahnaz Jamil-Copley demonstrated the system's effectiveness in a day-to-day clinical setting, completing five clinical cases during its first week of clinical use.
"We are excited to be CardioInsight's first commercial partner and I was very impressed with the ECVUE system's unique ability to display global cardiac activation without the need for an invasive study," said Dr. Prapa Kanagaratnam, the clinical lead for cardiac electrophysiology at St. Mary's Hospital, Imperial College Healthcare. "I think there is a real potential that this could be a disruptive technology that produces a paradigm shift in our approach to managing patients with cardiac arrhythmias."
St. Mary's Hospital has long been recognized as a leading institution in the study of cardiac electrophysiology, dating back to the first systematic clinical use of electrical recordings of the heart's activity in the early 1900's. Dr. Davies and his team later became a pioneering center for catheter-based 3D mapping.
"We are proud to have the opportunity to work with the world-class team from St. Mary's Hospital as we look to demonstrate the clinical impact of the ECVUE system," said Steve Arless, CEO of CardioInsight. "I believe their commitment to help pioneer our non-invasive mapping system is a testament to the ECVUE system's clinical potential, and we look forward to further demonstrating this potential under their guidance."
The commercial installation at St. Mary's Hospital is the first of a focused commercial introduction of the ECVUE system in Europe to demonstrate the unique ability to simplify cardiac mapping procedures, reduce procedure time, and expand the use of mapping to new applications.
For more information: www.cardioinsight.com


 September 05, 2025
September 05, 2025