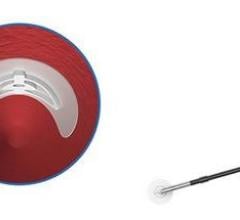
April 2, 2007 — Cordis Corp. has announced today the start of the pivotal trial for the ExoSeal Vascular Closure Device. The ECLIPSE Trial is a multicenter, non-blinded, randomized study designed to measure the safety and efficacy of the ExoSeal Vascular Closure Device versus manual compression to close vascular access sites in patients having undergone diagnostic or interventional procedures.
The trial will encompass 400 patients from 18 medical centers across the U.S.
On February 13, Principal Investigator Chiu Wong, M.D., associate professor of Medicine, New York-Presbyterian Hospital/Weill Cornell Medical Center and a Cordis Corp. Consultant, treated the first patient in the trial. Prior to the start of the ECLIPSE Trial, the ExoSeal(TM) Vascular Closure Device had been used in 150 patients participating in a first-in-human study for the device.
"The results from the first-in-man study were very encouraging, and this trial will help us determine whether those results can be maintained in a larger number of patients," said Dr. Wong. "As an interventional cardiologist, I welcome the opportunity to investigate a device that could help patients recover faster from a catheterization procedure."
The ExoSeal Vascular Closure Device features a synthetic bioabsorbable polymer and is being studied to determine whether it can enable expedited hemostasis (the cessation of bleeding), faster patient ambulation (ability to walk) and reduced bed-stay after a catheterization procedure. It also represents Cordis' entry into the vascular closure device market. Nearly eight million patients undergo cardiac catheterization procedures annually.
For more information visit www.cordis.com.

 October 07, 2025
October 07, 2025