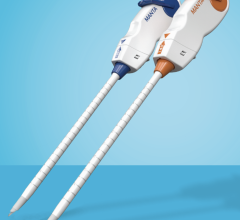
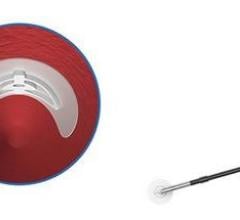
April 20, 2012 — VasoStitch will be featuring its technology at EuroPCR, Europe’s largest interventional cardiology scientific meeting, May 17, in Paris. The company is developing a percutaneous access-and-closure system for the nonsurgical deployment of large-diameter transcatheter therapeutic devices, such as transcatheter aortic valve implants (TAVI) and endovascular aneurysm repair (EVAR) devices. This system is designed to reduce procedural complexity and eliminate the need for open surgery.
The challenge with percutaneous catheter-based cardiac procedures is that a surgical cut-down is required for access and then closure. VasoStitch has developed two platform technologies designed to access and close percutaneous therapy sites without the need for open surgery, thus making catheter-based interventions simpler and more accessible.
“At the present time, surgical means of arterial or cardiac access-and-closure for large-diameter, catheter-based interventions such as TAVI adds about 60 minutes to an already lengthy and elaborate procedure. As a result, a device that can duplicate the current ‘gold standard’ of surgical access and repair — but in a quicker and simpler, nonsurgical procedure — will surely stimulate the adoption of large-diameter cardiovascular and endovascular therapies, as well as improve the efficiency of patient management,” said David W. J. Smith, president and CEO of VasoStitch. “We are moving quickly toward our first-in-human milestone.”
The VasoStitch presentation at EuroPCR will be made by the company’s founder, Amir Belson, M.D.
For more information: www.vasostitch.com

 October 07, 2025
October 07, 2025