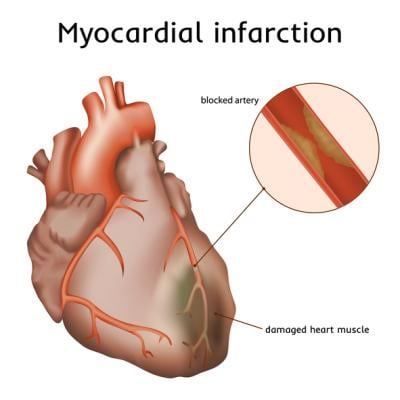
Heart attack (myocardial infarction), illustration. A blocked coronary artery has led to heart muscle damage. Getty Images
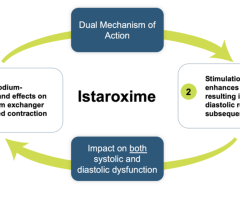
June 1, 2022 — Recardio's Phase 2 trial results demonstrated the excellent safety profile of its lead drug Dutogliptin along with data suggesting potential positive effects on cardiac function and were presented in May at the World Congress on Acute Heart Failure 2022 in Madrid. Along with data suggesting potential positive effects on cardiac function, it was concluded that a pivotal study of this therapeutic approach in a large, adequately powered NDA-enabling study is warranted.
After reviewing the Phase 2 trial results, the FDA agreed with the proposed trial design and sample size for the pivotal Phase 3 trial of Dutogliptin in co-adminstration with filgrastim in early recovery post-myocardial infarction. A comparable agreement was also reached with the EMA.
The global pivotal trial, named HEAL-MI, will enroll 4,000 acute myocardial infarction patients with STEMI but not specified left ventricular ejection fraction (LVEF) and with facilitated screening procedures, which will accelerate the total enrollment. The primary endpoint is a combined clinical endpoint with a Quality-of-Life evaluation as a secondary endpoint.
Based on this advice, preparations are already under way for the trial that is intended to start in the course of 2022 and will enroll patients in the key global regions - US, Europe and China.
Dr. Roman Schenk, Recardio President commented: "We appreciate the positive agency feedback and are looking forward to contributing with this HEAL-MI pivotal trial to further therapeutic options for the respective patients with myocardial infarction."
For more information: www.recardio.eu

 April 28, 2023
April 28, 2023